Back to C4 Home
C4: Quiz 5 – Answers
C4: Quiz 5 – Answers
1) metal + water –> metal hydroxide + hydrogen
2)
a) sodium + water –> sodium hydroxide + hydrogen
b) 2 Na + 2 H2O –> 2 NaOH + H2
3)
a) magnesium + water –> magnesium hydroxide + hydrogen
b)
i) 2+ or +2 or Mg2+
ii) 1- or -1 or - or OH-
iii) Mg(OH)2
c) Mg + 2 H2O –> Mg(OH)2 + H2
4) metal + acid –> salt + hydrogen
5)
a) magnesium + hydrochloric acid –> magnesium chloride + hydrogen
b) Mg + 2 HCl –> MgCl2 + H2
6)
a) zinc + sulfuric acid –> zinc sulfate + hydrogen
b) Zn + H2SO4 –> ZnSO4 + H2
7)
a) Hydrogen
b)
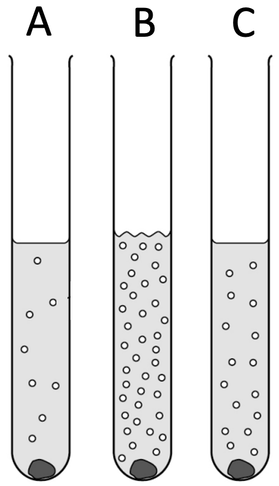
i) B is the most reactive metal as it has the greatest rate of reaction. We can tell that B has the greatest rate of reaction because it produces the greatest number of gas bubbles/ the greatest quantity of hydrogen
ii) B, C, A
c) Any 3 from:
e)
i) Temperature change
ii) The student would observe a greater temperature change for a more reactive metal
7)
a) Hydrogen
b)
i) B is the most reactive metal as it has the greatest rate of reaction. We can tell that B has the greatest rate of reaction because it produces the greatest number of gas bubbles/ the greatest quantity of hydrogen
ii) B, C, A
c) Any 3 from:
- The same mass of the metals
- The same surface area/ shape for the metals
- The same concentration of acid
- The same volume of acid
- The same initial temperature for the acid and metals
e)
i) Temperature change
ii) The student would observe a greater temperature change for a more reactive metal
Questions
1) Complete the word equation below for the reaction between a metal and water.
1) Complete the word equation below for the reaction between a metal and water.
2) A student reacts sodium with water.
a) Write the word equation for this reaction.
a) Write the balanced chemical equation for this reaction.
3) A student reacts magnesium with water.
a) Write the word equation for this reaction.
b)
i) Magnesium gives electron(s) to become a magnesium ion; use the periodic table to determine the charge on the magnesium ion.
ii) What is the charge on a hydroxide ion?
iii) Hence or otherwise, determine the chemical formula for magnesium hydroxide.
c) Write the balanced chemical equation for this reaction.
4) What is the general word equation for the reaction between a metal and an acid.
5) A student reacts magnesium with hydrochloric acid.
a) Write the word equation for this reaction.
b) Write the balanced chemical equation for this reaction.
6) A student reacts zinc with sulfuric acid.
a) Write the word equation for this reaction.
b) Write the balanced chemical equation for this reaction.
7) A student has 3 unknown metals called A, B and C. He places these 3 metals into different test tubes that all contain acid. The results of the reactions are shown below.
a) All of the reactions between the metals and acids produce a gas; what is the name of the gas produced?
b)
i) Which metal is the most reactive? Explain your answer.
ii) Put the 3 metals in order of reactivity starting from the most reactive to the least reactive.
c) When the student is completing this experiment, give 3 variables that they need to keep constant. You do not need to explain your answer.
d) Counting bubbles isn’t the most accurate way of investigating the rate of reaction. How could the student modify the experiment to measure the volume of gas produced more accurately?
e)
i) Give another variable that the student could have measured to work out the rate of reaction. Do not say anything about measuring the volume of gas.
ii) For your variable in part e i, say what the student would observe for a more reactive metal.
b)
i) Which metal is the most reactive? Explain your answer.
ii) Put the 3 metals in order of reactivity starting from the most reactive to the least reactive.
c) When the student is completing this experiment, give 3 variables that they need to keep constant. You do not need to explain your answer.
d) Counting bubbles isn’t the most accurate way of investigating the rate of reaction. How could the student modify the experiment to measure the volume of gas produced more accurately?
e)
i) Give another variable that the student could have measured to work out the rate of reaction. Do not say anything about measuring the volume of gas.
ii) For your variable in part e i, say what the student would observe for a more reactive metal.