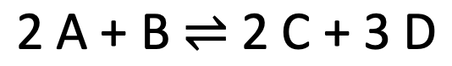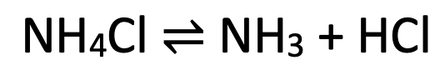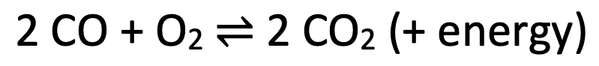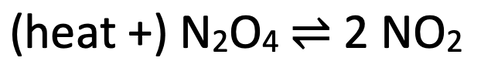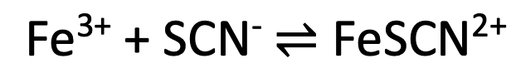C6: Quiz 7 – Answers
1)
a) A dynamic equilibrium is where there are the same number of forwards and backwards reactions taking place. This results in the quantities/ concentrations of all of the substances remaining constant
b) closed system/ sealed container
c) Yes – reactions are still taking place
2) Temperature, pressure and concentration
3)
a) decreasing, endothermic, exothermic
If we increase the temperature, the reversible reaction will respond by decreasing the temperature. This means that there will be more endothermic reactions, and fewer exothermic reactions.
b) decreasing, fewer
If we increase the pressure, the reversible reaction will respond by decreasing the pressure. This moves the position of the equilibrium towards the side of the reversible reaction with fewer molecules/ moles of gas.
c) products
If we increase the concentration of one of the reactants, the reversible reaction will respond by making more of the products.
4)
a) exothermic
b) The system will respond to the higher temperature by decreasing the temperature. It does this by doing more endothermic reactions, which is the forwards reaction. This means that the position of the dynamic equilibrium will move towards the right where we have a greater concentration of C & D and a lower concentration of A & B
c) The system will respond to the higher pressure by decreasing the pressure. This favours the side with fewer molecules/ moles of gas. The left side has 3 molecules and the right side has 5 molecules. Therefore, an increase in pressure means that there will be more backwards reactions. This moves the position of the dynamic equilibrium to the left (the side with fewer gas molecules), which means that we will have more A & B and less C & D
d) The reversible reaction will respond to a decrease in C & D by reversing the change and increasing the concentration of C & D. This happens because there will be more forwards reactions taking place as there is lots of A & B, and fewer backwards reactions taking place as there is little C & D. So, a decrease in the concentration of C & D moves the position of the dynamic equilibrium to the right.
5)
a) Forwards is endothermic, and backwards is exothermic
b) The system will respond to the lower temperature by increasing the temperature. It can do this by doing more exothermic reactions, which is the backwards reaction. This means that the position of the dynamic equilibrium will move towards the left where we have a greater concentration of ammonia chloride (the left), and a lower concentration of ammonia and hydrogen chloride (the right)
c) The system will respond to a decrease in pressure by increasing the pressure. This favours the side with more molecules/ moles of gas. The left side has 1 molecule and the right side has 2 molecules. Therefore, a decrease in pressure means that there will be more forwards reactions. This moves the position of the dynamic equilibrium to the right (the side with more gas molecules), which means that we will have a greater concentration of ammonia and hydrogen chloride (the right), and a lower concentration of ammonia chloride (the left).
6)
a) Low temperature. The forwards reaction is exothermic as energy is given out. Therefore, we can push the position of the dynamic equilibrium to the right by having a low temperature as a low temperature favours the exothermic reaction, which means more forwards reactions and therefore a greater concentration of carbon dioxide. A low temperature moves the position of the dynamic equilibrium to the right.
b) High pressure. There are 3 molecules/ moles of gas on the left and 2 molecules/ moles of gas on the right. As there are less molecules of gas on the right (the side that we want), we should have a high pressure as a higher pressure favours the side with fewer molecules/ moles of gas. A high pressure moves the position of the dynamic equilibrium to the right.
7)
a)
i) Darker brown
ii) Lighter brown
b) Lighter brown. For this reaction, the forwards reaction is endothermic as energy is required, and the backwards reaction is exothermic. A decrease in temperature favours the exothermic reaction as the system will want to respond by producing more temperature; the system will want to reverse the decrease in temperature. This means that we have more exothermic reactions (the backwards reaction), which pushes the dynamic equilibrium to the left where we have an increased concentration of dinitrogen tetroxide (N2O4) and a decreased concentration of nitrogen dioxide (NO2). Nitrogen dioxide (NO2) is brown, so a lower concentration of nitrogen dioxide will mean that the colour of the equilibrium mixture will become a lighter brown.
c) Lighter brown. For this reaction, there is 1 molecule/ mole of gas on the left, and 2 molecules/ moles of gas on the right. An increase in pressure favours the side with fewer molecules of gas as the system will want to respond by decreasing the pressure; the system will want to reverse the increase in pressure. The left side has fewer molecules of gas, so an increase in pressure pushes the position of the dynamic equilibrium to the left. This means that we will have more backwards reactions, which increases the concentration of dinitrogen tetroxide (N2O4) and decreases the concentration of nitrogen dioxide (NO2). Nitrogen dioxide is brown, so a lower concentration of nitrogen dioxide will mean that the colour of the equilibrium mixture will become a lighter brown.
8)
a) Yellow
b) Reddish-brown
c)
i) As soon as the iron (III) ions are added, the concentration of iron (III) ions will increase, which will result in the solution becoming more yellow
ii) The system will respond to the increase in the concentration of iron (III) ions by reducing the concentration of iron (III) ions; reversing the change. This means that there will be more forwards reactions and less backwards reactions. This will result in the concentration of iron thiocyanate ions increasing, thus meaning that the solution will become more reddish-brown.
a) What is a dynamic equilibrium?
b) Fill in the gap in the following sentence; a dynamic equilibrium can only occur in a __________ __________?
c) Are reactions still taking place at a dynamic equilibrium; yes or no?
2) What are the 3 factors that affect the position of a dynamic equilibrium?
3) Le Chatelier says that if we change any of the 3 factors that are the answer to question 2, the reversible reaction will respond by counteracting the change.
a) Fill in the gaps below with either “increasing”, “decreasing”, “endothermic” or “exothermic”.
If we increase the temperature, the reversible reaction will respond by _______ the temperature. This means that there will be more _______ reactions, and fewer _______ reactions.
b) Fill in the gaps below with either “increasing”, “decreasing”, “fewer” or “more”.
If we increase the pressure, the reversible reaction will respond by _______ the pressure. This moves the position of the equilibrium towards the side of the reversible reaction with _______ molecules/ moles of gas.
c) Fill in the gap below with either “reactants” or “products”.
If we increase the concentration of one of the reactants, the reversible reaction will respond by making more of the _______.
4) We have the reversible reaction below.
a) Is the backwards reaction endothermic or exothermic?
b) We now increase the temperature of the system. In which direction will the position of the dynamic equilibrium move? Explain your answer.
c) We now increase the pressure of the system. In which direction will the position of the dynamic equilibrium move? Explain your answer.
d) We now decrease the concentration of C & D in the system. In which direction will the position of the dynamic equilibrium move? Explain your answer.
5) When we heat ammonium chloride, it breaksdown into ammonia and hydrogen chloride. This reaction is a reversible reaction and it is taking place in a closed container. The reversible reaction is:
b) We now decrease the temperature of the system. In which direction will the position of the dynamic equilibrium move? Explain your answer.
c) We now decrease the pressure of the system. In which direction will the position of the dynamic equilibrium move? Explain your answer.
6) The reaction of carbon monoxide and oxygen to produce carbon dioxide is a reversible reaction. The reversible reaction is shown below.
I want to move the dynamic equilibrium as far towards the right as possible; I want as much carbon dioxide as possible.
a) Should I use a low or high temperature? Explain your answer.
b) Should I use a low or high pressure? Explain your answer.
7) Dinitrogen tetroxide (N2O4) can decompose to produce nitrogen dioxide (NO2). The reversible reaction for this is shown below.
Dinitrogen tetroxide (N2O4) is a colourless gas and nitrogen dioxide (NO2) is a brown gas.
a) Assume that the dynamic equilibrium is in the middle.
i) If the dynamic equilibrium moved towards the right (more NO2), how would you expect the colour of the equilibrium mixture to change; would it become lighter brown or darker brown?
ii) If the dynamic equilibrium moved towards the left (more N2O4), how would you expect the colour of the equilibrium mixture to change; would it become lighter brown or darker brown?
b) We are now going to decrease the temperature of the vessel that the reaction is taking place in. Predict the colour change of the equilibrium mixture; would it become lighter brown or darker brown? Explain your answer.
c) We are now going to increase the pressure of the vessel that the reaction is taking place in. Predict the colour change of the equilibrium mixture; would it become lighter brown or darker brown? Explain your answer.
8) Iron (III) ions (Fe3+) can react with thiocyanate ions (SCN-) to produce iron thiocyanate ions (FeSCN2+). All 3 of these substances are in a solution (aqueous). This reaction is reversable and it is shown below.
Iron (III) ions are yellow, thiocyanate ions are colourless and iron thiocyanate ions are reddish-brown. The reaction is going to take place in a closed system.
a) If the position of the dynamic equilibrium was on the left, what would the colour of the equilibrium mix be?
b) If the position of the dynamic equilibrium was on the right, what would the colour of the equilibrium mix be?
c) A student increases the concentration of iron (III) ions (Fe3+).
i) Describe the colour change immediately after the iron (III) ions (Fe3+) are added.
ii) Describe the colour change when the new dynamic equilibrium has been reached. Explain your answer.