1) A hydrocarbon is a compound that is made out of hydrogen and carbon only
(make sure that you say only)
2) Covalent bonds – covalent bonds are where two non-metals share pairs of electrons with each other; there is then a strong force of attraction between the positively charged nuclei of the atoms and the shared pair(s) of electrons
3) An alkane is a hydrocarbon that contains only single (carbon-carbon) bonds
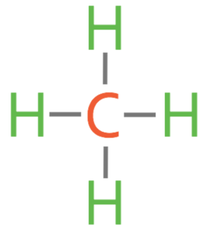
4)
5) CnH2n+2
6)
Methane: CH4
Ethane: C2H6
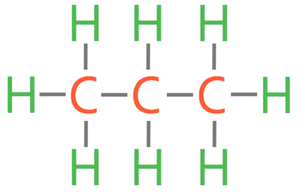
Propane: C3H8
Butane: C4H10
7)
8)
a) C7H16
b) C15H32
c) C23H48
d) C31 H64
9)
a) Yes – alkane
b) No – not an alkane
c) No – not an alkane
d) Yes – alkane
10) Longer hydrocarbons have higher melting & boiling points because they have stronger intermolecular forces of attraction. These stronger intermolecular forces of attraction require more energy to overcome, thus meaning that longer hydrocarbons have greater melting and boiling points.
11)
a) Flammability refers to how easy it is to ignite a substance (ignite means set fire to). The more flammable something is, the easier it is to set fire to it
b) Shorter hydrocarbons are more flammable
12)
a) Viscosity refers to how easily a liquid (or gas) flows. The more viscous something is, the less easily it flows (golden syrup). The less viscous something is, the more easily it flows (water)
b) Golden syrup
c) Longer hydrocarbons
13)
a) Volatility refers to how easy a substance will turn into a gas/ evaporate
b) Shorter hydrocarbons are more volatile
14)
a) B because it is a longer alkane, which means that it has stronger intermolecular forces of attraction that require more energy to break, thus meaning that B has a higher melting & boiling point
b) A because it is a shorter hydrocarbon
c) B because it is a longer hydrocarbon
1) What is a hydrocarbon?
2) What bonds are involved in hydrocarbons?
3) What is an alkane?
4) The first alkane is methane (CH4). Draw the displayed formula for methane.
5) What is the general molecular formula for alkanes (the formula with n in it)?
6) Write down the names and molecular formulas for the first 4 alkanes.
7) Draw the displayed formula for propane.
8)
a) What would be the molecular formula for an alkane with 7 carbons in?
b) What would be the molecular formula for an alkane with 15 carbons in?
c) What would be the molecular formula for an alkane with 23 carbons in?
d) What would be the molecular formula for an alkane with 31 carbons in?
9) Say whether the hydrocarbons below are alkanes.
a) C5H12
b) C12H24
c) C19H36
d) C24H50
10) Do shorter or longer hydrocarbons have higher melting & boiling points? Explain your answer.
11)
a) What does flammability mean?
b) Are shorter or longer hydrocarbons more flammable?
12)
a) What does viscosity mean?
b) Out of golden syrup and water, which is more viscous?
c) Out of shorter and longer hydrocarbons, which are more viscous?
13)
a) What does volatility mean?
b) Which are more volatile out of shorter and longer hydrocarbons?
14) We have the two alkanes below:
> Alkane A – C10H22
> Alkane B – C40H82
a) Which alkane will have the higher melting & boiling point? Explain your answer.
b) Which alkane is less viscous? Explain your answer.
c) Which alkane is less flammable? Explain your answer.