1) A hydrocarbon is a compound that is made out of hydrogen and carbon only
Make sure that you say only
2) Alkanes are known as saturated hydrocarbons as all of the bonds in the alkane are single covalent bonds; alkanes only contain single (carbon-carbon) bonds. Also, we cannot fit any more hydrogens onto alkanes
3) Alkenes are hydrocarbons that contain a double carbon-carbon bond
4) CnH2n
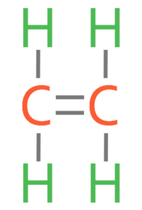
5)
6)
a) C3H6
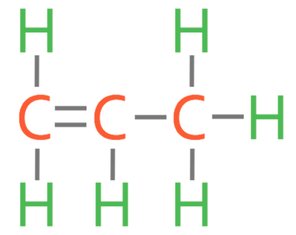
b)
7)
a) Yes – alkene
b) No – not an alkene
c) No – not an alkene
d) Yes – alkene
8) The steps are:
> Add a few drops of the hydrocarbon into a test tube containing bromine water (which is orange)
> Shake the test tube
- If the hydrocarbon is an alkane, no reaction will take place and the solution in the test tube will remain orange
- If the hydrocarbon is an alkene, the bromine will react with the alkene, which results in the solution going from orange to colourless
9) Alkenes are more reactive
10) hydrocarbon + oxygen –> carbon dioxide + water
Or
hydrocarbon + oxygen –> carbon dioxide + water (+ energy)
11) C8H16 + 12 O2 –> 8 CO2 + 8 H2O
1) What is a hydrocarbon?
2) Why are alkanes known as saturated hydrocarbons?
3) What is an alkene?
4) Write down the general molecular formula for an alkene.
5) The first alkene is ethene (C2H4). Draw the displayed formula for ethene.
6) Propene contains 3 carbon atoms.
a) What is the molecular formula for propene?
b) Draw the displayed formula for propene.
7) Say whether the following hydrocarbons are alkenes.
a) C5H10
b) C3H8
c) C11H24
d) C14H28
8) How can we test if a hydrocarbon is an alkene or an alkane?
9) Are alkanes or alkenes more reactive?
10) Write the word equation for the complete combustion of a hydrocarbon.
11) Write the balanced chemical equation for the combustion of the alkene C8H16.