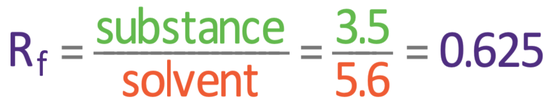C8 B) Paper Chromatography
- The mobile phase – the molecules that can move. This is always a liquid or a gas. For paper chromatography with water, the mobile phase is the water that moves up the filter paper carrying different substances in the sample with it.
- The stationary phase – the molecules that cannot move. This is a solid or thick liquid. For paper chromatography with water, the stationary phase is the filter paper.
When a chromatography experiment is taking place, the different substances in the sample are in the two different phases in different proportions. The amount of time that the different substances spend in each of the phases is dependent upon the solubility of the substances in the solvent (the mobile phase), and the attractiveness of the substances to the filter paper (the attractiveness to the solid or thick liquid in the stationary phase); these 2 factors vary for different substances, solvents and the solids/ thick liquids used in the stationary phase.
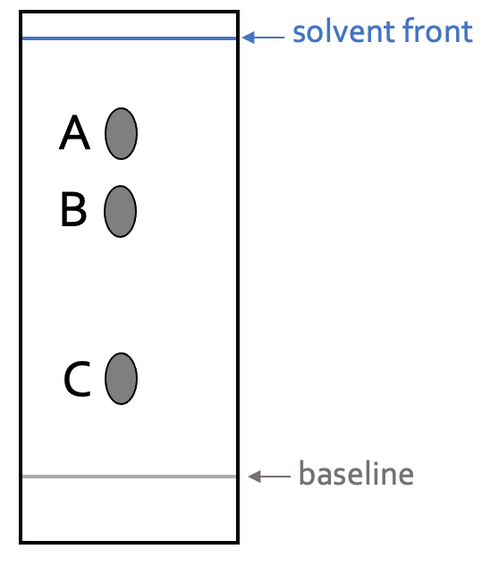
The different proportions of time spent in the mobile and stationary phase is why the different substances in the sample move different distances up the filter paper – this is why we get spots at different heights on the chromatogram. An example of a chromatogram for an ink on filter paper is shown below.
A substance that is more soluble in the solvent and less attracted to the filter paper will spend longer in the mobile phase compared to the stationary phase. This means that the substance will move higher up the filter paper – out of the 3 different substances that the ink is made out of, A spends the longest time in the mobile phase compared to the stationary phase, hence why it is the highest spot.
A substance that is less soluble in the solvent and more attracted to the filter paper will spend longer in the stationary phase compared to the mobile phase. This means that the substance will move a shorter distance up the filter paper – out of the 3 different substances that the ink is made out of, C spends the longest time in the stationary phase compared to the mobile phase, hence why it is the lowest spot.
The order and distances of the spots on a chromatogram may change depending on the solvent that is used and the chromatography paper. Examples of different solvents are water and ethanol. This is because different substances have different solubilities in different solvents.
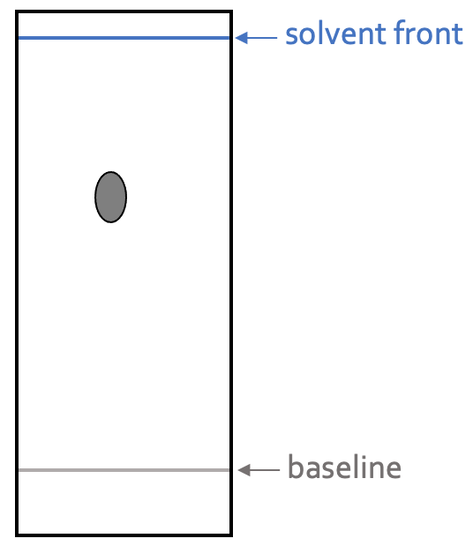
A pure substance is a substance that only contains one single element or one type of molecule. A pure substance will always only produce one spot on the chromatogram irrespective of the solvent and chromatography paper used. This is because the pure substance only contains one substance. Mixtures (or impure substances) will produce multiple spots on the chromatogram. An example of a chromatogram of a pure substance is shown below.
Working Out the Rf value
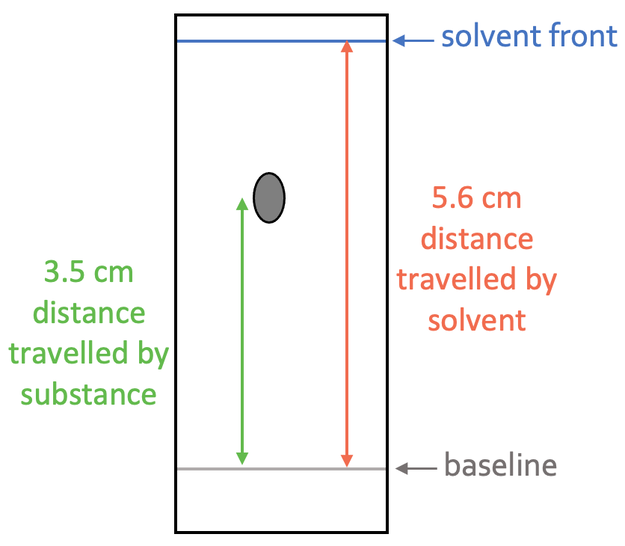
The Rf value tells us the ratio of the distance travelled by the substances and the distance travelled by the solvent. We work out the Rf value by using the formula below:
We work out the Rf value by subbing the values into the formula above. The “distance travelled by substance” is 3.5 cm, and the “distance travelled by solvent” is 5.6 cm; we sub these values into the formula:
The Rf value for the substance on the chromatogram is 0.625.
The Rf value for substances depends on the solvent that we use. If we use a solvent that the substance is more soluble in, the substance in question will spend more time in the mobile phase and less time in the stationary phase, which will mean that the substance will move further up the chromatogram and therefore have a value for Rf that is closer to 1.
If we use a solvent that the substance is less soluble in, the substance in question will spend more time in the stationary phase and less time in the mobile phase, which will mean that the substance will move a smaller distance up the chromatogram and therefore have a value for Rf that is closer to 0.
We are able to obtain a more accurate Rf value by using a longer piece of chromatography paper.
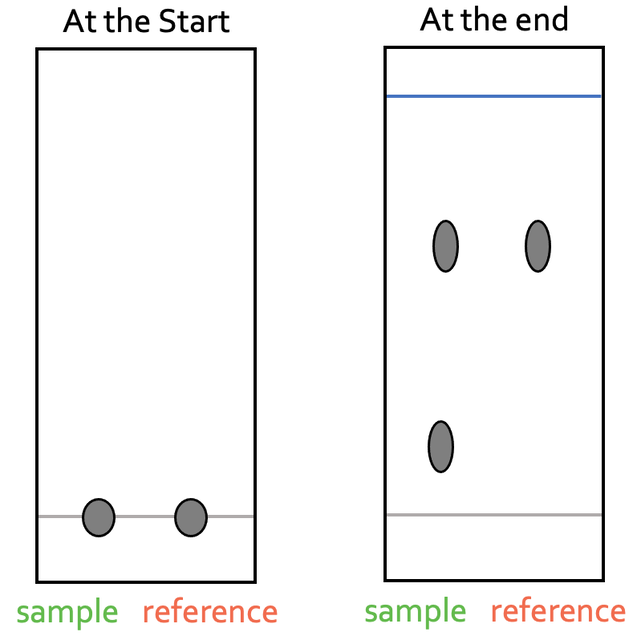
We can use chromatography to check whether a mixture contains a certain substance. We are able to do this by placing a sample of the mixture that we are testing on the baseline. We then place a sample of the pure substance that we are testing for on the baseline next to the sample that we are testing. The pure substance will be called the reference substance. We then undertake chromatography in the normal way and compare the spots on the chromatogram. The chromatogram at the start and the end of the chromatography experiment for the presence of a pure substance is shown below.
From the above chromatogram, we can see that the sample is a mixture as the sample produces two spots on the chromatogram. We can also see that one of the spots in the sample is level with the reference substance. We work out the Rf values for any spot in the mixture that is around the same height as the reference. If the Rf values are the same, it tells us that the substance that we are testing for (the reference substance) may be present in the mixture.
In order to be certain that the substance that we are testing for is definitely present in our sample, we need to use a variety of different solvents and check whether the Rf values are the same in the different solvents. We use a variety of different solvents because different substances have different solubilities in different solvents. If the Rf values are the same in all of the different solvents, it is very likely that the reference substance is present in the mixture. If the Rf values are only the same for some of the solvents, it means that the reference substance is not present in the mixture.