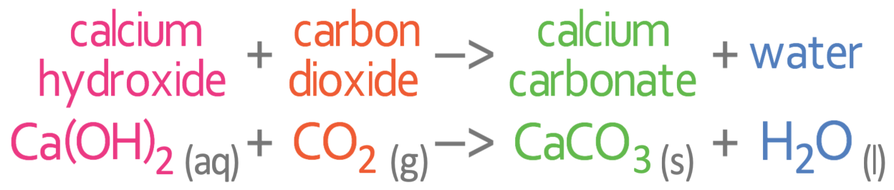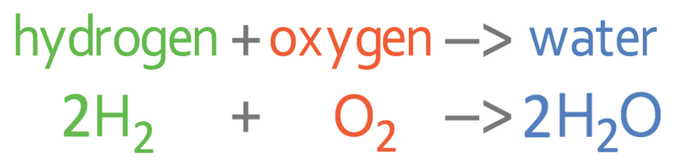Back to C8 Home
C8 C) Test for Gases
C8 C) Test for Gases
There are a variety of different tests for different gases. We need to be careful when testing for gases because some of the gases produced from reactions can be dangerous. For example, the gases could be flammable or poisonous. If the gases produced from a reaction are flammable, positions or you are unsure what the products of a reaction are, you should complete the experiment under a fume cupboard. The fume cupboard will take away any harmful gases which protects the individual carrying out the experiment and protects other individuals in a laboratory.
Carbon Dioxide
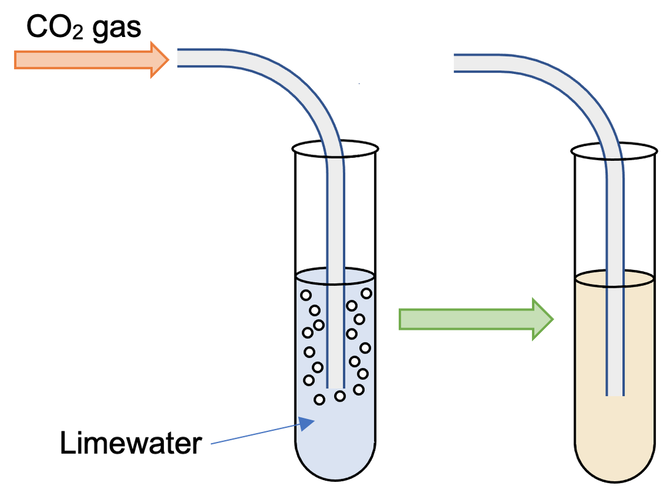
We use limewater to test for the presence of carbon dioxide. Limewater is calcium hydroxide dissolved in water. To test for carbon dioxide, we pump the gas that may contain carbon dioxide through the limewater. If carbon dioxide is present in the gas, the limewater will turn cloudy (milky).
We use limewater to test for the presence of carbon dioxide. Limewater is calcium hydroxide dissolved in water. To test for carbon dioxide, we pump the gas that may contain carbon dioxide through the limewater. If carbon dioxide is present in the gas, the limewater will turn cloudy (milky).
The cloudiness is caused by the calcium hydroxide reacting with the carbon dioxide to form calcium carbonate; the calcium carbonate is insoluble in the water and is therefore a solid (it is a precipitate). Water is also produced when calcium hydroxide and carbon dioxide react. The equation for the reaction of calcium hydroxide and carbon dioxide is:
If carbon dioxide is not present in the gas, the limewater will remain colourless.
Hydrogen
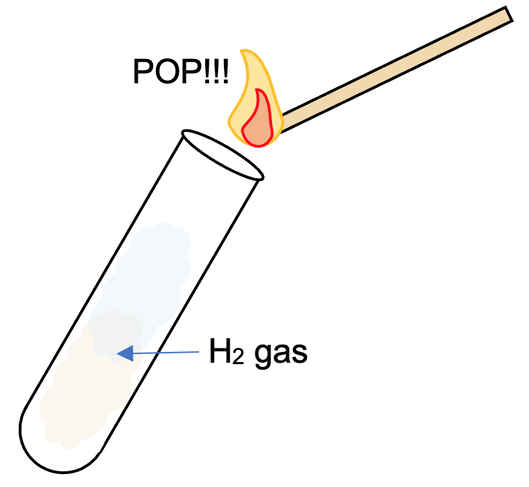
We can test for the presence of hydrogen gas by holding a lit splint above where the hydrogen gas may be (e.g. a test tube). If hydrogen is present, we will hear a ‘squeaky pop’ sound.
We can test for the presence of hydrogen gas by holding a lit splint above where the hydrogen gas may be (e.g. a test tube). If hydrogen is present, we will hear a ‘squeaky pop’ sound.
The ‘squeaky pop’ sound is created because the hydrogen gas is burnt quickly in the oxygen in air. The equation for hydrogen reacting with oxygen is:
If hydrogen is not present, we will not hear a ‘squeaky pop’ sound.
Oxygen
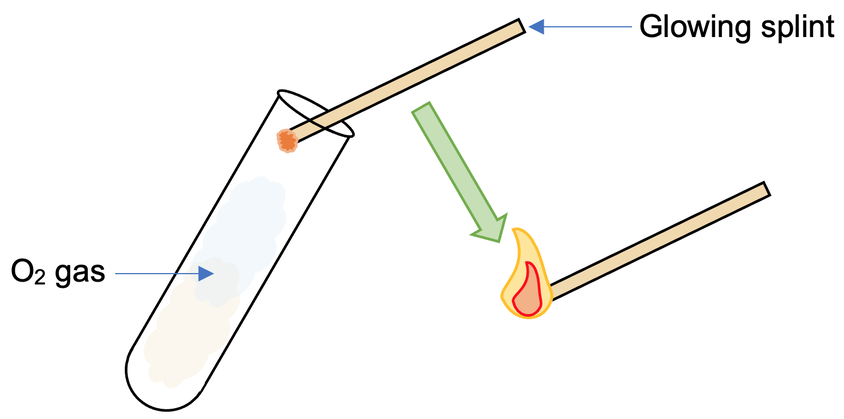
We can test for the presence of oxygen by placing a glowing splint (which is created by blowing out a lit splint) above where the oxygen may be (e.g. a test tube). If oxygen is present, the glowing splint will relight. If oxygen is not present, the glowing splint will not relight.
We can test for the presence of oxygen by placing a glowing splint (which is created by blowing out a lit splint) above where the oxygen may be (e.g. a test tube). If oxygen is present, the glowing splint will relight. If oxygen is not present, the glowing splint will not relight.
Chlorine
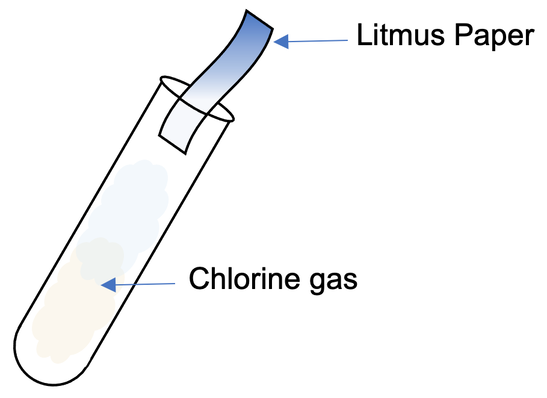
We can test for the presence of chlorine by using damp blue litmus paper. When damp blue litmus paper is placed over chlorine gas, the chlorine will bleach the litmus paper, resulting in the litmus paper becoming white. The litmus paper may turn red before it turns white because the solution of chlorine is acidic.
We can test for the presence of chlorine by using damp blue litmus paper. When damp blue litmus paper is placed over chlorine gas, the chlorine will bleach the litmus paper, resulting in the litmus paper becoming white. The litmus paper may turn red before it turns white because the solution of chlorine is acidic.