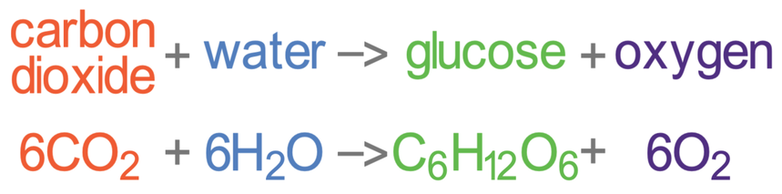Back to C9 Home
C9 A) Evolution of the Earth’s Atmosphere
C9 A) Evolution of the Earth’s Atmosphere
For the last 200 million years, the composition of the atmosphere has been about the same as it is today with about 80% being nitrogen, 20% being oxygen and a small proportion being other gases such as carbon dioxide, water vapour and the noble gases. However, the atmosphere has been different to this in the past.
Scientists cannot be certain as to what happened in the earth’s early atmosphere as it is hard to gather evidence from billions of years ago. This has led to many different theories as to how the earth’s atmosphere has changed over time. Here is one of those theories.
Scientists cannot be certain as to what happened in the earth’s early atmosphere as it is hard to gather evidence from billions of years ago. This has led to many different theories as to how the earth’s atmosphere has changed over time. Here is one of those theories.
The Early Atmosphere
For the first billion years of the earth’s existence, there was intense volcanic activity leading to an atmosphere that was mostly carbon dioxide, some water vapour and a little bit of methane and ammonia. There would have been essentially no oxygen. The atmosphere would have been very much like the atmospheres of Mars and Venus today. The volcanic activity will have also released nitrogen, which would have led to the levels of nitrogen in the atmosphere increasing over time. The levels of nitrogen would have increased over time because nitrogen is very unreactive.
For the first billion years of the earth’s existence, there was intense volcanic activity leading to an atmosphere that was mostly carbon dioxide, some water vapour and a little bit of methane and ammonia. There would have been essentially no oxygen. The atmosphere would have been very much like the atmospheres of Mars and Venus today. The volcanic activity will have also released nitrogen, which would have led to the levels of nitrogen in the atmosphere increasing over time. The levels of nitrogen would have increased over time because nitrogen is very unreactive.
Moving to our atmosphere now
There are 2 main changes from the early atmosphere to the atmosphere of today. One of the changes is that carbon dioxide concentrations decreased because of photosynthesis and carbon dioxide being dissolved in the oceans. The second change is that the concentration of oxygen increased because of photosynthesis. Here is a bit more detail on both of these changes.
Decreasing Carbon Dioxide
As the earth cooled down, the water vapour in the atmosphere condensed, which resulted in oceans forming.
Most of the carbon dioxide in the atmosphere dissolved into the oceans. The carbon in the oceans reacted to form carbonate precipitates resulting in sediments on the seabed.
Around 2.7 billion years ago, algae evolved and started to photosynthesis. Photosynthesis takes carbon dioxide from the atmosphere and releases oxygen. Photosynthesis caused the level of carbon dioxide in the atmosphere to fall and the level of oxygen to rise.
Marine animals started to evolve, and their shells and skeletons contained some of the carbonates that were present in the ocean.
When the plants, plankton and animals died, they fell to the bottom of the seabed and got buried by layers of sediment (mud). Over millions of years, heat and pressure turned the dead material into fossil fuels (coal, crude oil and natural gas) or limestone.
There are 2 main changes from the early atmosphere to the atmosphere of today. One of the changes is that carbon dioxide concentrations decreased because of photosynthesis and carbon dioxide being dissolved in the oceans. The second change is that the concentration of oxygen increased because of photosynthesis. Here is a bit more detail on both of these changes.
Decreasing Carbon Dioxide
As the earth cooled down, the water vapour in the atmosphere condensed, which resulted in oceans forming.
Most of the carbon dioxide in the atmosphere dissolved into the oceans. The carbon in the oceans reacted to form carbonate precipitates resulting in sediments on the seabed.
Around 2.7 billion years ago, algae evolved and started to photosynthesis. Photosynthesis takes carbon dioxide from the atmosphere and releases oxygen. Photosynthesis caused the level of carbon dioxide in the atmosphere to fall and the level of oxygen to rise.
Marine animals started to evolve, and their shells and skeletons contained some of the carbonates that were present in the ocean.
When the plants, plankton and animals died, they fell to the bottom of the seabed and got buried by layers of sediment (mud). Over millions of years, heat and pressure turned the dead material into fossil fuels (coal, crude oil and natural gas) or limestone.
- Coal is a fossil fuel which is formed from dead trees. The dead trees were buried by flooding, which prevented oxidation of the wood taking place due to a lack of oxygen. Over millions of years of compression and heat, the dead trees turned into coal.
- Crude oil and natural gas are made from deposits of plankton that have died and sunk to the bottom of the sea or the bottom of lakes. The plankton was then buried by layers of sediment, which prevented oxidation of the dead material due to a lack of oxygen. Over millions of years, the dead plankton has become crude oil and natural gas.
- Limestone is formed from shells and skeletons of marine organisms.
Increasing Oxygen
Over time, other plants evolved. These plants and algae photosynthesised. The equation for photosynthesis is shown below:
Over time, other plants evolved. These plants and algae photosynthesised. The equation for photosynthesis is shown below:
Photosynthesis takes carbon dioxide out of the atmosphere, so the levels of carbon dioxide decreased. Photosynthesis produces oxygen, which resulted in the levels of oxygen in the atmosphere increasing.
As the concentration of oxygen in the atmosphere increased, it allowed more complex life (like animals) to evolve.
Eventually, around 200 million years ago, the atmosphere reached the composition that it is today with around 80% being nitrogen, 20% being oxygen and a small proportion being other gases such as carbon dioxide, water vapour and the noble gases.
As the concentration of oxygen in the atmosphere increased, it allowed more complex life (like animals) to evolve.
Eventually, around 200 million years ago, the atmosphere reached the composition that it is today with around 80% being nitrogen, 20% being oxygen and a small proportion being other gases such as carbon dioxide, water vapour and the noble gases.