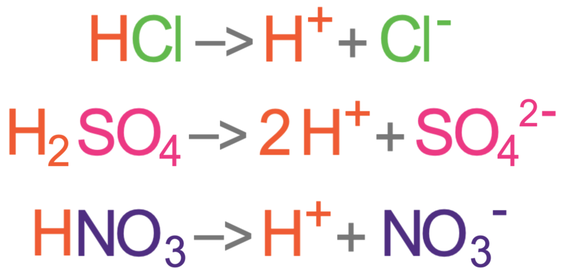C4 C) Strong & Weak Acids
Acids ionise in aqueous solutions to produce positive hydrogen ions (H+ ions). For example, hydrochloric acid (HCl) ionises in water to produce H+ ions and negative chloride ions (Cl-). The equation for this is shown below:
On the left of the above reaction we have the undissociated acid, and on the right of the above reaction we have the dissociated acid.
Another example is nitric acid (HNO3) that ionises in water to produce H+ ions and negative nitrate ions (NO3-). The equation for this is shown below:
Strong & Weak Acids
Strong acids completely ionise when they are dissolved in water to produce H+ ions. Another way of saying this is that all of the acid particles dissociate fully to produce H+ ions. Examples of strong acids are hydrochloric acid (HCl), sulfuric acid (H2SO4) and nitric acid (HNO3). The equations for these acids are shown below.
Weaker acids do not fully ionise when they are dissolved in water to produce H+ ions. Instead, only a small proportion of the acid particles dissociate to produce H+ ions. The ionisation of a weak acid is a reversible reaction (a reaction that can go in both directions). An example of a weak acid is ethanoic acid and the equation for ethanoic acid ionising is shown below.
The sign in the middle tells us that the reaction is reversible. All reversible reactions reach a dynamic equilibrium whereby the concentrations of all of the substances in the reaction remain constant. Therefore, there will be an equilibrium between the undissociated acid (the left) and the dissociated acid (right). For a weak acid, few of the acid particles will dissociate to produce H+ ions – the equilibrium will be towards the left.
The pH of an acid is determined by the concentration of H+ ions. This means that for the same acid concentration, a stronger acid will have a greater concentration of H+ ions compared to a weaker acid – this is because a stronger acid will fully dissociate to produce H+ ions, and a weaker acid will only partially dissociate which produces fewer H+ ions. Therefore, for the same acid concentration, a stronger acid will have a lower pH than a weaker acid. The number of H+ ions determines how quickly acids react. Stronger acids produce a greater quantity of H+ ions (for the same acid concentration), which means that stronger acids will react faster than weaker acids.
Strong & Concentrated
The strength of an acid tells us what proportion of the acid particles dissociate to become H+ ions. In a strong acid, all of the acid particles dissociate to produce H+ ions. In a weak acid, only some of the acid particles dissociate to produce H+ ions.
The concentration of an acid refers to the number of acid particles that we have in a given volume. The formula triangle for concentration is shown below.
A more concentrated acid will have more acid particles in a given volume, which means that there will be more H+ ions, thus resulting in a lower pH (remember, the number of H+ ions determines the pH of an acid – a lower pH has more H+ ions). This is true for both strong and weak acids.
Let’s now suppose that we have two acids with the same pH, and that one of the acids is made out of a strong acid and the other is made out of a weak acid. In order for these two acids to have the same pH, the number of H+ ions would need to be the same. As weaker acids only partially dissociate in the solution to produce H+ ions, we would need to have a greater concentration of the weaker acid in the solution to produce the same quantity of H+ ions, thus meaning that the pH of the two acids are the same (the alternative way of viewing this is that the concentration of the stronger acid would be lower than the concentration of the weaker acid because the stronger acid fully dissociates to produce H+ ions).
Questions
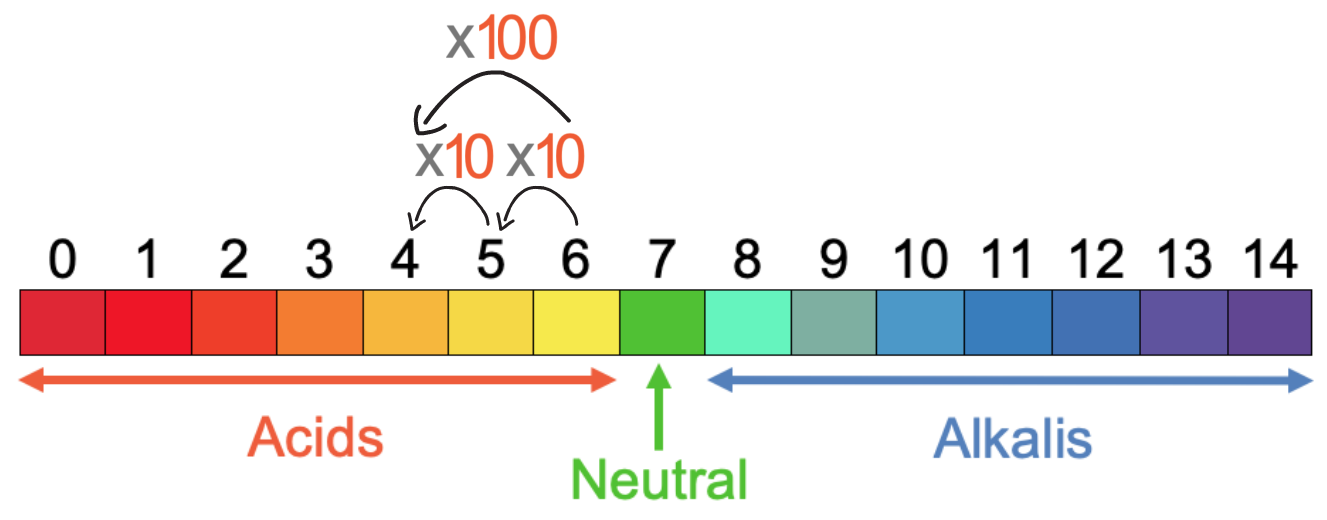
We are now going to have a look at a few questions to do with concentration and pH. In the previous section, we learnt that as we move down the pH values for acids, the concentration of H+ ions increases by a factor of 10. So, there are 10 times as many H+ ions for a pH of 5 compared to a pH of 6. And there are 10 times as many H+ ions for a pH of 4 compared to a pH of 5. This means that there are 100 times as many H+ ions in a solution with a pH of 4 compared with a solution with a pH of 6; to go from a pH of 6 to a pH of 4, we multiply the number of H+ ions by 10 two times, which means that the number of H+ ions increases by 100.
We can be asked to work out the pH of an acid if the concentration of the acid changes. We are now going to have two examples.
Example 1
An acid has a concentration of 2 g/dm3 and a pH of 3. Find the pH if the acid is diluted resulting in the concentration becoming 0.2 g/dm3.
We find the pH of the diluted acid by seeing how the concentration of the acid has changed. The original concentration was 2 g/dm3 and the final concentration is 0.2 g/dm3. This means that the acid concentration is now 10 times less. As the concentration is 10 times less, the concentration of H+ ions will also be 10 times less. This means that the pH of the acid will increase by 1 from 3 to 4. The pH of the acid after it has been diluted is 4.
These questions can be a little bit tricky to get your head around. Therefore, it is always worth looking at your answer and checking that it makes sense/ we have moved the pH in the correct direction. The question told us that the acid was diluted, and we know that the pH of an acid that is more dilute is closer to neutral (a pH of 7). The pH for our answer was 4, which is closer to 7 from the original pH of 3. This implies that we have completed the working correctly.
Example 2
An acid has a concentration of 3 g/dm3 and a pH of 1.4. The acid is then diluted which results in it having a concentration of 0.03 g/dm3. Find the pH of the diluted acid.
Like before, we find the pH of the diluted acid by seeing how the concentration has changed. The original concentration was 3 g/dm3 and the new concentration is 0.03 g/dm3. This means that the acid concentration is now 100 times less, which means that the concentration of H+ ions will also be 100 times less. An increase by 1 in pH for acids represents a decrease in the concentration of H+ ions by 10 times. We are decreasing the concentration of H+ ions by 100, which means that the pH of the acid increases by 2. The original pH of the acid was 1.4 and an increase in pH by 2 will result in the pH of the diluted acid being 3.4.
We should check that this makes sense, which it does because a diluted acid will have a higher pH than a more concentrated acid.
The answer is that the pH of the final diluted acid is 3.4.