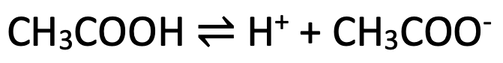C4: Quiz 3 – Answers
1) H+ ions
2) HCl –> H+ + Cl-
3) HNO3 –> H+ + NO3-
4) Strong acids completely ionise/ dissociate when dissolved in water to produce H+ ions
Weak acids do not fully ionise/ dissociate when dissolved in water to produce H+ ions
5) Hydrochloric acid would have the lower pH because it is a stronger acid, which means that it will fully dissociate to produce H+ ions in a solution (ethanoic acid is a weaker acid, so it will only partially dissociate to produce H+ ions). This means that hydrochloric acid will have a greater concentration of H+ ions, thus meaning that it has a lower pH value.
6)
a) The reaction is reversible
b) A dynamic equilibrium is where the concentrations of all of the substances remains constant; the concentration of the undissociated acid (the left) and dissociated acid (right) remains constant
c) For the same concentration, a stronger acid will produce more H+ ions as it will fully ionise/ dissociate when it dissolves in water (compared to the ethanoic acid that will only partially ionise/ dissociate). As the stronger acid produces more H+ ions, it will have a lower pH value than the ethanoic acid
7) a factor of 10
8) 5
9) 4
10) 4.6
11) 6.1
1) What ion do acids form when they dissolve in water?
2) Hydrochloric acid is HCl. Write the dissociation equation for HCl dissociating to produce a H+ ion.
3) Nitric acid is HNO3. Write the dissociation equation for HNO3 dissociating to produce a H+ ion.
4) What is the difference between a strong acid and a weak acid?
5) Hydrochloric acid is a strong acid and ethanoic acid is a weak acid. If we had two beakers of the two acids with the same volume and concentration, which acid would have the lower pH? Explain your answer.
6) Ethanoic acid is a weak acid. The equation for the dissociation is shown below.
a) What does the symbol in the middle mean?
b) This dissociation will reach a dynamic equilibrium; what does dynamic equilibrium mean?
c) For the same concentration, will a stronger acid produce more H+ ions than ethanoic acid? Will the pH of the stronger acid be lower or higher than the pH of ethanoic acid? Explain your answer.
7) As we go down the pH scale by 1 for acids (e.g. from 4 to 3), by what factor does the concentration of H+ ions increase by?
8) An acid has a concentration of 9 g/dm3 and a pH of 4. The acid is diluted resulting in it having a concentration of 0.9 g/dm3. What is the pH of the diluted acid?
9) An acid has a concentration of 25 g/dm3 and a pH of 2. The acid is diluted resulting in it having a concentration of 0.25 g/dm3. What is the pH of the diluted acid?
10) An acid has a concentration of 3 mol/dm3 and a pH of 1.6. The acid is diluted resulting in it having a concentration of 0.003 mol/dm3. What is the pH of the diluted acid?
11) An acid has a pH of 2.1 and a concentration of 5 mol/dm3. I add some water to the acid, which results in the concentration becoming 0.0005 mol/dm3. Find the pH of the solution after I have added some water.