C5 D) Investigating Energy Transfer
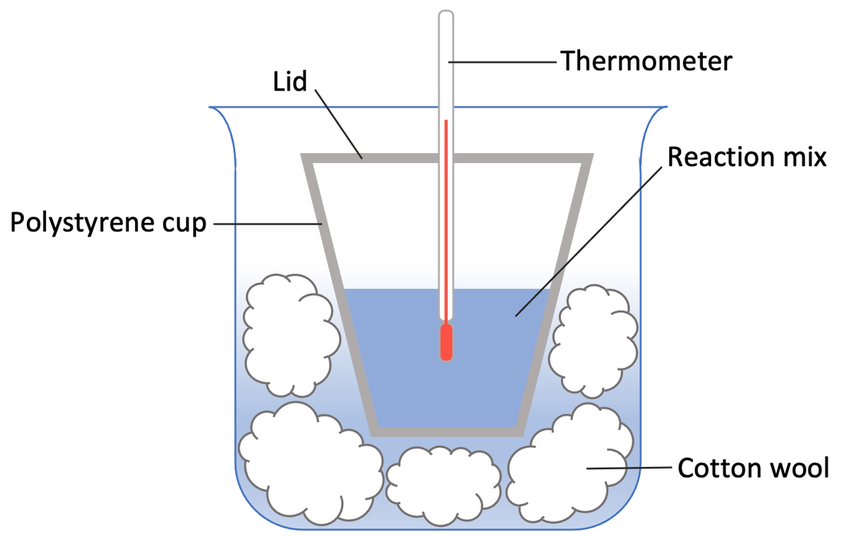
There is a lot of insulation in this experiment and this is to reduce the amount of energy that is lost to the environment. Example of insulation in this experiment are the polystyrene cup with a lid, and the polystyrene cup being placed inside a beaker full of cotton wool.
In addition to using this experiment to see whether the reaction is endothermic or exothermic, we can also use the experiment to see how different factors affect the temperature change of a reaction. For example, we can modify the concentrations of the reactants (e.g. a more concentrated acid), change the surface area to volume ratio of the reactants (e.g. use powdered reactants), change the mass of the reactants etc….
We are now going to look at how we would investigate the effect that the concentration of hydrochloric acid has on the temperature change when hydrochloric acid reacts with sodium hydroxide. In order to investigate this, we would undertake the following steps:
1) Put a certain volume (like 25 cm3) of 0.25 mol/dm3 of hydrochloric acid and sodium hydroxide in different beakers (the volume and concentration of sodium hydroxide will be the same every time).
2) We then place both of the separate beakers in a water bath and heat them up to a certain temperature (like 30°C).
3) When both of the beakers are at the desired temperature (30°C), we mix them together in the polystyrene cup and place the lid on.
4) We then monitor the temperature and record the highest temperature.
5) We then repeat all of the previous steps for some different concentrations of hydrochloric acid; we would undertake the same steps for a concentration of 0.5 mol/dm3, a concentration of 0.75 mol/dm3, a concentration of 1 mol/dm3 etc.
6) We then compare our results for temperature change against concentration.
The neutralisation reaction of hydrochloric acid and sodium hydroxide is an exothermic reaction; it gives out energy. As we increase the concentration of hydrochloric acid, we should observe that there is a greater change in temperature. This is because as we increase the concentration of hydrochloric acid, there are more hydrochloric acid particles in the same volume of the reaction mixture, which means that there will be more frequent collisions, thus meaning that there will be a greater rate of reaction and therefore a greater temperature increase.