C9 D) Air Pollution
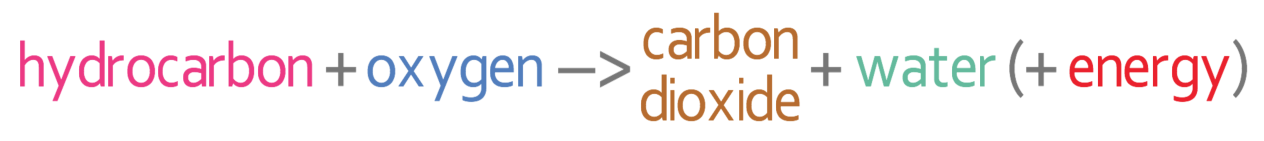
However, if there is not enough oxygen present, the hydrocarbon may undergo incomplete combustion. Incomplete combustion can result in solid particles (particulates) of soot (carbon) and unburnt fuel being released. Also, incomplete combustion can result in carbon monoxide (CO) rather than carbon dioxide being produced (CO2).
We are now going to have a look at the issues with particulates and carbon monoxide.
One issue with particulates is that they can be inhaled and damage the respiratory system (the lungs and trachea).
The particulates that are produced during incomplete combustion enter the atmosphere where they can cause an effect that is known as global dimming. Global dimming is the gradual reduction of sunlight reaching the earth’s surface due to particulates in the atmosphere from human activity. Global dimming happens because the particulates in the atmosphere absorb and reflect sunlight back into space, thus meaning that less light reaches the earth’s surface.
You can see the presents of soot (carbon) from incomplete combustion by looking at the top of a large glass candle. When most of the candle has been burnt, you will start to notice that the top of the glass will start to go black and this is because of the soot (carbon) that is released during incomplete combustion.
Carbon monoxide (CO) affects the body’s ability to take oxygen around the body. Red blood cells are responsible for taking oxygen around the body. Red blood cells contain haemoglobin. In the lungs, haemoglobin combines with oxygen to create oxyhaemoglobin, and then the oxygen is taken around the body. If carbon monoxide is taken into the lungs, the haemoglobin will bind with the carbon monoxide instead of the oxygen because carbon monoxide is more reactive. This means that less of the haemoglobin binds with the oxygen, so your body transports less oxygen around the body. Oxygen is essential as we need it for respiration reactions. A lack of oxygen being transported around the body can result in an individual fainting, become unconscious and maybe even dying.
Carbon monoxide is an odourless (has no smell) and colourless gas. This means that it is really hard to detect, which makes it even more dangerous! You can purchase carbon monoxide detectors for your house which will detect if carbon monoxide is present – check whether your house has one, and if it doesn’t, encourage your parents or guardians to purchase one!
Fossil fuels contain sulphur impurities. When fossil fuels are burnt, the sulphur impurities react with oxygen to create sulfur dioxide (SO2).
Also, the temperature inside engines that burn fossil fuels is quite high, which provides the energy needed for nitrogen and oxygen in air to react to form nitrogen oxides (NOX).
The sulfur dioxide and nitrogen oxides produced mix with water in clouds to produce dilute sulfuric acid and dilute nitric acid, which then falls as acid rain. There are a variety of effects of acid rain, and here are some of them:
- Acidification of lakes or rivers – acid rain falling in rivers or lakes makes them acidic. Some bacteria, fish, plants and aphids will be unable to survive in the more acidic conditions, which can result in them dying. This can have a huge effect on ecosystems because ecosystems are very interconnected, so the death of one species will impact the whole food web. This can result in ecosystems changing.
- Acidification of soil – acid rains results in soil that the rain lands on becoming more acidic, which can cause trees and plants to die. This can also have an effect of the whole ecosystem.
- Dissolving of buildings – acid rain dissolves buildings that are made out of limestone, sandstone or marble. This is because the acid rain reacts with these rocks. Below is a picture of a sculpture that has been affected by acid rain.
- Respiratory issues – sulfur dioxide and nitrogen oxides can cause respiratory issues if they are breathed in.