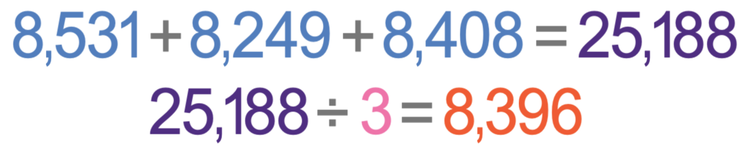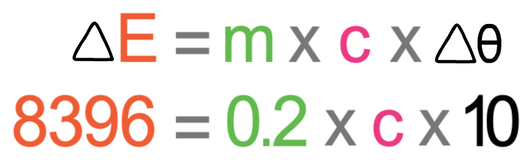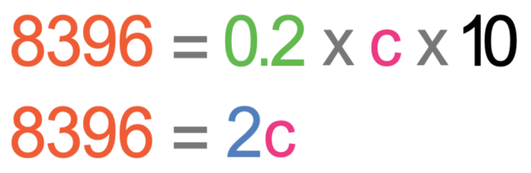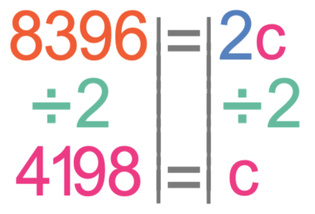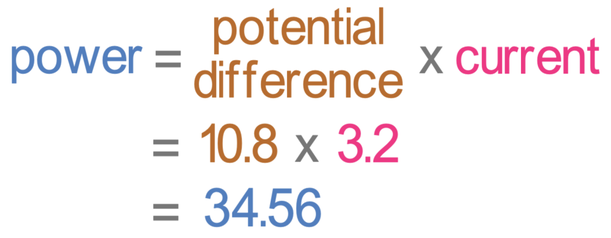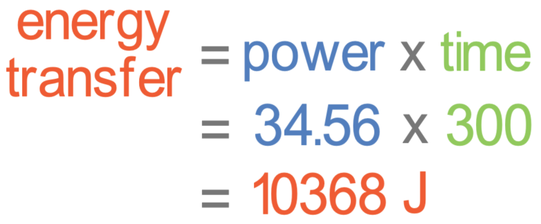Back to P1 Home
P1 F) Investigating Specific Heat Capacity
P1 F) Investigating Specific Heat Capacity
We learnt in the previous section that the formula for working out the change in energy in the thermal energy store of a substance can be worked out by using the formula below.
Whereby:
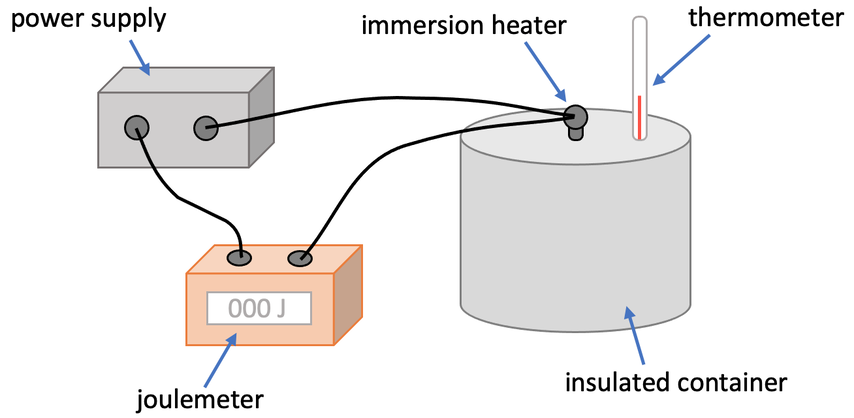
We are now going to look at an experiment that we can undertake to find the specific heat capacity of a particular substance (either a solid or a liquid). In order to work out the specific heat capacity of a substance (c), we need to know the change in thermal energy (ΔE), the mass (m) and the change in temperature (Δθ). We set up the apparatus for the experiment like what is shown below.
- ΔE is the change in thermal energy in joules (J)
- m is the mass of the substance in kg
- c is the specific heat capacity in J/kg°C
- Δθ is the change in temperature in °C
We are now going to look at an experiment that we can undertake to find the specific heat capacity of a particular substance (either a solid or a liquid). In order to work out the specific heat capacity of a substance (c), we need to know the change in thermal energy (ΔE), the mass (m) and the change in temperature (Δθ). We set up the apparatus for the experiment like what is shown below.
We will be heating up the substance using an electric immersion heater. A joulemeter will be part of the circuit and it will measure the amount of energy used by the electric immersion heater. We assume that all of the energy from the immersion heater is transferred to the thermal energy stores of the substance that we are testing. Therefore, the reading on the joulemeter will be the value for change in thermal energy (ΔE).
The liquid or solid that we are finding the specific heat capacity of will be placed in the insulated container. The container is insulated to reduce the amount of heat that is lost to the surroundings (it reduces systemic error). This means that we obtain a more accurate value for change in thermal energy and therefore a more accurate value for specific heat capacity. Before we place our substance into the insulated container, we measure the mass by using a mass balance (a set of scales); this will give us the value for m in the formula.
Before we start the experiment, we need to find the initial temperature of the substance using the thermometer and ensure that the joulemeter is set to zero. The next step is to turn the immersion heater on, which causes the temperature of the substance to increase. We then wait for the temperature of the substance to increase by a certain amount, such as 10°C (a greater increase in temperature will give you a more accurate value for specific heat capacity). We then repeat the experiment a few times and work out the mean for the reading on the joulemeter (the change in thermal energy).
The next step is to place all of your values into the change in thermal energy formula and work out the specific heat capacity (c).
The liquid or solid that we are finding the specific heat capacity of will be placed in the insulated container. The container is insulated to reduce the amount of heat that is lost to the surroundings (it reduces systemic error). This means that we obtain a more accurate value for change in thermal energy and therefore a more accurate value for specific heat capacity. Before we place our substance into the insulated container, we measure the mass by using a mass balance (a set of scales); this will give us the value for m in the formula.
Before we start the experiment, we need to find the initial temperature of the substance using the thermometer and ensure that the joulemeter is set to zero. The next step is to turn the immersion heater on, which causes the temperature of the substance to increase. We then wait for the temperature of the substance to increase by a certain amount, such as 10°C (a greater increase in temperature will give you a more accurate value for specific heat capacity). We then repeat the experiment a few times and work out the mean for the reading on the joulemeter (the change in thermal energy).
The next step is to place all of your values into the change in thermal energy formula and work out the specific heat capacity (c).
Example 1
Let’s suppose that we carried out this experiment to find the specific heat capacity of water. We measured the mass as 200 grams and the temperature change as 10°C. We completed the experiment 3 times and the readings for the joulemeter for the 3 experiments were 8 531 j, 8 249 j and 8 408 j. Find the specific heat capacity of water.
The easiest way to find the specific heat capacity of water is to sub all of the values from the question into the change in thermal energy formula. This formula is shown below.
Let’s suppose that we carried out this experiment to find the specific heat capacity of water. We measured the mass as 200 grams and the temperature change as 10°C. We completed the experiment 3 times and the readings for the joulemeter for the 3 experiments were 8 531 j, 8 249 j and 8 408 j. Find the specific heat capacity of water.
The easiest way to find the specific heat capacity of water is to sub all of the values from the question into the change in thermal energy formula. This formula is shown below.
We are looking for the specific heat capacity of water, which is the value of c in the above formula.
The change in thermal energy (ΔE) for the water is the readings on the joulemeter. We completed the experiment 3 times, so we need to find the mean of the 3 readings. We find the mean by adding the 3 values up and then we divide by 3. The working is shown below.
The change in thermal energy (ΔE) for the water is the readings on the joulemeter. We completed the experiment 3 times, so we need to find the mean of the 3 readings. We find the mean by adding the 3 values up and then we divide by 3. The working is shown below.
The mean change in thermal energy (ΔE) is 8 396 joules.
We now need the other values in the formula. m in the formula is the mass, which is measured in kilograms. We are told in the question that the mass of water is 200 grams. 1 kg is 1,000 g, so we convert grams to kilograms by dividing by 1,000; the mass is 0.2 kg (200 ÷ 1,000 = 0.2).
Δθ in the formula is the change in temperature (Δθ). The question tells us that the change in temperature is 10°C.
We sub all of these values into the formula; ΔE is 8 396 j, m is 0.2 kg and Δθ is 10°C.
We now need the other values in the formula. m in the formula is the mass, which is measured in kilograms. We are told in the question that the mass of water is 200 grams. 1 kg is 1,000 g, so we convert grams to kilograms by dividing by 1,000; the mass is 0.2 kg (200 ÷ 1,000 = 0.2).
Δθ in the formula is the change in temperature (Δθ). The question tells us that the change in temperature is 10°C.
We sub all of these values into the formula; ΔE is 8 396 j, m is 0.2 kg and Δθ is 10°C.
We now solve this equation in the same way that we would solve an algebraic equation to find the value of an unknown. The first step in solving the equation is to multiply the two numbers on the right side of the equation; 0.2 x 10.
We want to find the value of c and not 2c. Therefore, we divide both sides of the equation by 2.
This tells us that the specific heat capacity of water is 4 198 j/kg°C.
Not Using a Joulemeter
The experiment at the top of this section had a joulemeter that measured the amount of energy that was transferred to the material (the change in thermal energy). The reading on the joulemeter was the exact value for the energy that was transferred.
You can also work out the energy transferred without a joulemeter if you find the potential difference of the immersion heater (measured by a voltmeter), the current (measured by an ammeter) and the time that the immersion heater is on for. The first step is to find the power of the immersion heater, which is the rate of energy transfer. We work out power by multiplying the potential difference by current:
The experiment at the top of this section had a joulemeter that measured the amount of energy that was transferred to the material (the change in thermal energy). The reading on the joulemeter was the exact value for the energy that was transferred.
You can also work out the energy transferred without a joulemeter if you find the potential difference of the immersion heater (measured by a voltmeter), the current (measured by an ammeter) and the time that the immersion heater is on for. The first step is to find the power of the immersion heater, which is the rate of energy transfer. We work out power by multiplying the potential difference by current:
Power is in watts and 1 watt is 1 joule of energy per second.
After we have the power of the immersion heater, we can then work out energy transferred by multiplying the power by the time (in seconds).
After we have the power of the immersion heater, we can then work out energy transferred by multiplying the power by the time (in seconds).
Let’s have an example.
Example 2
We heat up some magnesium for 5 minutes. The current is 3.2 A and the potential difference is 10.8 V. Find the energy transferred.
The first step in finding the energy transferred is to find the power, which we do by multiplying the potential difference (10.8 V) by the current (3.2 A).
Example 2
We heat up some magnesium for 5 minutes. The current is 3.2 A and the potential difference is 10.8 V. Find the energy transferred.
The first step in finding the energy transferred is to find the power, which we do by multiplying the potential difference (10.8 V) by the current (3.2 A).
The power is 34.56 watts.
We can now find the energy transferred by multiplying the power (34.56) by time. The time needs to be in seconds. The question tells us that the time is 5 minutes. We convert minutes to seconds by multiplying by 60; the time in seconds is 300 seconds (5 x 60 = 300). We can now find the energy transferred; the power is 34.56 watts and the time is 300 seconds.
We can now find the energy transferred by multiplying the power (34.56) by time. The time needs to be in seconds. The question tells us that the time is 5 minutes. We convert minutes to seconds by multiplying by 60; the time in seconds is 300 seconds (5 x 60 = 300). We can now find the energy transferred; the power is 34.56 watts and the time is 300 seconds.
The energy transferred is 10,368 joules.
If the question gave us the mass and change in temperature, we could work out the specific heat capacity by subbing the energy transferred (10,368 joules), the mass and change in temperature into the change in thermal energy formula.
Note
You can combine the power and energy transferred formula to get just one formula to work out energy transferred. The formula comes about because we replace “power” with “potential difference multiplied by current”. The working for the formula is shown below:
If the question gave us the mass and change in temperature, we could work out the specific heat capacity by subbing the energy transferred (10,368 joules), the mass and change in temperature into the change in thermal energy formula.
Note
You can combine the power and energy transferred formula to get just one formula to work out energy transferred. The formula comes about because we replace “power” with “potential difference multiplied by current”. The working for the formula is shown below:
I think that it is best to remember the two separate formulas but feel free to just remember this one.