Back to P1 Home
P1 H) Reducing Unwanted Energy Transfer
P1 H) Reducing Unwanted Energy Transfer
Energy can never be created or destroyed. Instead, energy is transferred usefully, stored or dissipated.
A useful device transfers energy from one energy store to another energy store that is useful. But not all of the inputted energy is transferred to the useful energy store because some of the energy will be dissipated (wasted). The dissipated energy is usually thermal energy; the useful device and the surroundings get hot. For example, a phone transfers energy from the chemical energy store of the battery to the useful energy stores of the phone. However, some of the inputted energy will be dissipated/ transferred to energy stores that are not useful like the thermal energy stores of the phone (the phone will get hot which is not useful).
Let’s look at another example of a petrol-powered car. The engine of a petrol-powered car transfers energy from the chemical energy store of the petrol to the kinetic energy store of the car. However, not all of the inputted energy is transferred to the kinetic energy store of the car, and instead some of the energy is dissipated; some of the energy will be transferred to the thermal energy stores of the car and the surroundings; the car/ engine and the surroundings get hot.
A useful device transfers energy from one energy store to another energy store that is useful. But not all of the inputted energy is transferred to the useful energy store because some of the energy will be dissipated (wasted). The dissipated energy is usually thermal energy; the useful device and the surroundings get hot. For example, a phone transfers energy from the chemical energy store of the battery to the useful energy stores of the phone. However, some of the inputted energy will be dissipated/ transferred to energy stores that are not useful like the thermal energy stores of the phone (the phone will get hot which is not useful).
Let’s look at another example of a petrol-powered car. The engine of a petrol-powered car transfers energy from the chemical energy store of the petrol to the kinetic energy store of the car. However, not all of the inputted energy is transferred to the kinetic energy store of the car, and instead some of the energy is dissipated; some of the energy will be transferred to the thermal energy stores of the car and the surroundings; the car/ engine and the surroundings get hot.
Reducing Unwanted Energy Transfer
There are a few different ways that we can reduce unwanted energy transfer.
Lubricating
One way that we can reduce unwanted energy transfer is through lubrication. A frictional force is created when two or more components move past each other. This frictional force transfers energy from the kinetic energy stores of the moving parts to the thermal energy stores of the moving parts (the parts become hot). We can put a lubricant on moving parts, which reduces the amount of friction and therefore means that less energy is transferred to the thermal energy stores of the moving parts (less energy is dissipated/ wasted). An example of a lubricant is oil, which is used in engines and on the chains/ gears of bikes.
There are a few different ways that we can reduce unwanted energy transfer.
Lubricating
One way that we can reduce unwanted energy transfer is through lubrication. A frictional force is created when two or more components move past each other. This frictional force transfers energy from the kinetic energy stores of the moving parts to the thermal energy stores of the moving parts (the parts become hot). We can put a lubricant on moving parts, which reduces the amount of friction and therefore means that less energy is transferred to the thermal energy stores of the moving parts (less energy is dissipated/ wasted). An example of a lubricant is oil, which is used in engines and on the chains/ gears of bikes.
Reducing Conduction & Convection
We can reduce the rate of energy transfer from heating by choosing materials that have the characteristics that we want. Heat can be transferred from one place to another through conduction and convection.
Conduction
Conduction of thermal energy is where particles with more energy in their kinetic energy stores (hotter particles) pass some of their kinetic energy to neighbouring particles by vibrating and knocking into neighbouring particles. If we have a piece of metal that is warm at one end and cold at the other end, the particles at the warm end will pass on some of their kinetic energy to their neighbouring particles who then pass on energy to their neighbouring particles, which will eventually warm up the particles at the other end of the piece of mental (the end that was originally cold). Thermal conductivity measures how quick thermal energy is transferred through a material; a material with a high thermal conductivity (e.g. a metal) will transfer thermal energy at a faster rate than a material with a low thermal conductivity (e.g. polystyrene). We make products out of particular materials based on their thermal conductivity. For example, we make frying pans out of various metals because metals have a high thermal conductivity.
We can reduce the rate of energy transfer from heating by choosing materials that have the characteristics that we want. Heat can be transferred from one place to another through conduction and convection.
Conduction
Conduction of thermal energy is where particles with more energy in their kinetic energy stores (hotter particles) pass some of their kinetic energy to neighbouring particles by vibrating and knocking into neighbouring particles. If we have a piece of metal that is warm at one end and cold at the other end, the particles at the warm end will pass on some of their kinetic energy to their neighbouring particles who then pass on energy to their neighbouring particles, which will eventually warm up the particles at the other end of the piece of mental (the end that was originally cold). Thermal conductivity measures how quick thermal energy is transferred through a material; a material with a high thermal conductivity (e.g. a metal) will transfer thermal energy at a faster rate than a material with a low thermal conductivity (e.g. polystyrene). We make products out of particular materials based on their thermal conductivity. For example, we make frying pans out of various metals because metals have a high thermal conductivity.
Convection
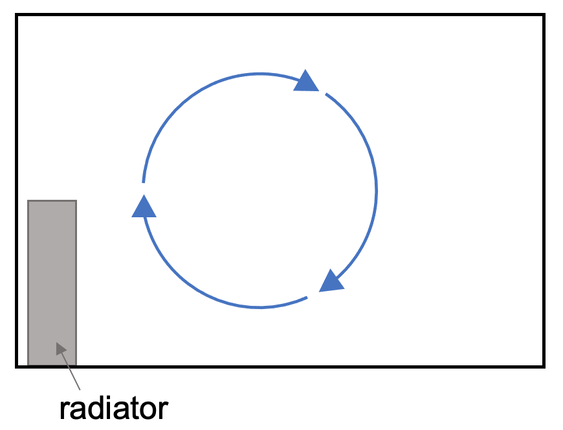
Heat energy can also be transferred by convection. Convection is when particles that have more energy in their kinetic energy stores (hotter particles) move around faster which increases the distances between the particles. This results in the density of the particles with more kinetic energy decreasing, which means that the particles with more kinetic energy (hotter particles) rise above denser cooler regions where the particles have less kinetic energy (colder particles); these hotter particles rise up and take their energy with them. Cooler particles then replace the hotter particles that have just risen, which creates convection currents.
The hotter particles that rose up then loose some of their kinetic energy (they cool down) and the distances between the particles decreases. This causes the density of the particles to increase, which results in the particles sinking. A cycle of warmer particles rising and cooler particles falling creates a convection current.
Convection only occurs in liquids and gases because the particles can move (convection does not occur in solids because the particles in solids are in fixed positions and therefore cannot move). There is a diagram below of a convection current in a room caused by a radiator.
House Design – Limiting Wasted Energy
The rate of heat loss from a building is affected by the thickness of its walls and the thermal conductivity of the materials used in the walls. There are many different ways that individuals can minimise the amount of heat that is lost from one’s home. There is an incentive for households to minimise the amount of heat lost because heating homes is very expensive. There are many methods to reduce the amount of heat that is lost from a home and some are more cost effective than others.
Here are some of the methods to reduce the energy transfer/ heat loss in a home:
- Loft insulation – this is a layer of fibreglass wool underneath the floorboards in the loft. The insulation has a low thermal conductivity, which means that there is very little conduction from the floor below the loft to the loft. This means that the floor in the loft is colder, which reduces convection currents in the loft. Loft insulation tends to be fairly cheap and is a good way to reduce heat loss in a home.
- Double glazing – double glazing is made out of 2 layers of glass with a gap of air in the middle. The gap of air reduces the amount of conduction through the window. Double glazing is fairly expensive.
- Curtains – windows are in contact with the outside and are considerably colder than the inside of the room. If there were no curtains, the windows would cause the temperature of the room to decrease because of conduction and convection. Curtains provide an air buffer against the cold glass in the windows, thus reducing the amount of heat loss through conduction. Curtains are cheap and provide a significant saving.
- Cavity wall insulation – the exterior walls of homes are made out of 2 walls with an air gap in between the two walls. The air gap reduces the amount of energy lost through conduction. We can reduce the amount of energy lost through convection in the cavity between the walls by filling the cavity with insulation. The insulation has a very low thermal conductivity, which means that less energy is transferred through the insulation and therefore less energy is lost. Cavity wall insulation is quite expensive.
- Draught proofing – this reduces heat loss through convection. Draught proofing is making sure that all draughts are prevented from coming inside the house. You can draught proof your front door. Draught proofing is cheap and an effective way to reduce energy loss.