Back to P1 Home
P1: Quiz 6
P1: Quiz 6
1) Write down the equation for working out the change in energy in a substance’s thermal energy stores. Write the units for all of the values as well.
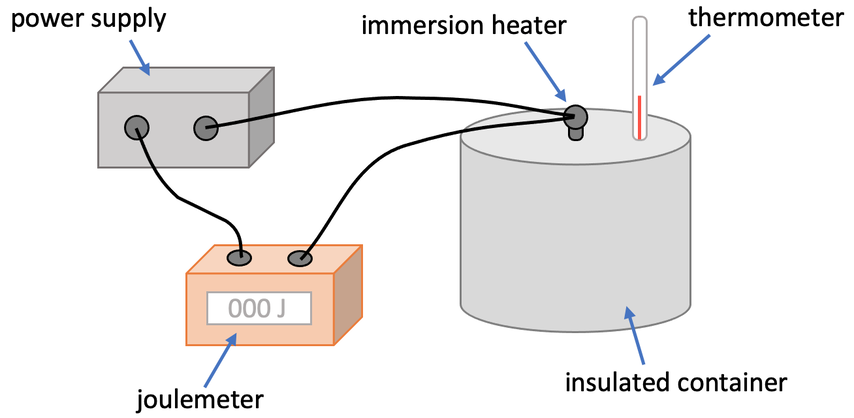
2) We can find the specific heat capacity of a liquid by using the apparatus shown below.
2) We can find the specific heat capacity of a liquid by using the apparatus shown below.
a) What does the joulemeter measure with respect to the specific heat capacity formula?
b) Why do we use an insulated container with a lid?
c) Mass appears in the specific heat capacity formula. How can we measure the mass of the liquid?
d) What piece of apparatus do we use to measure the change in temperature of the liquid?
3) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the unknown liquid was 300 grams, the change in temperature was 25°C and the reading on the joulemeter for change in energy was 9,750 joules. Work out the specific heat capacity of this liquid.
4) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the liquid is 150 grams. Before the immersion heater was switched on, the temperature of the liquid was 21°C.
The student leaves the immersion heater on until it transfers 15 kilojoules of energy to the liquid. After the immersion heater has transferred 15 kilojoules of energy to the liquid, the temperature of the liquid is 63°C.
Find the specific heat capacity of the liquid. Give your answer to 2 significant figures.
5) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the liquid is 350 grams. The student increases the temperature of the liquid by 20°C. She completes her experiment 3 times and the readings on the joulemeter are 13,032 J, 12,856 J and 12,962 J.
a) Find the mean energy transfer from the 3 experiments.
b) Find out the specific heat capacity of this liquid.
6) A student undertakes an experiment to find the specific heat capacity of marble. They use all of the apparatus in question 2 except a joulemeter. They put 300 grams of marble into the insulated container and turn the immersion heater on for 6 minutes. During these 6 minutes, the temperature of the marble increases by 55°C. The current in the circuit is 4.5 A and the potential difference is 9 V.
a) Find the power of the immersion heater.
b) Find the energy transferred by the immersion heater.
c) Find the specific heat capacity of marble. Give your answer to 2 significant figures.
7) A student undertakes an experiment to find the specific heat capacity of polypropylene. They use all of the apparatus in question 2 except a joulemeter. They put 170 grams of polypropylene into the insulated container. The initial temperature of the polypropylene is 18°C. The immersion heater is on for 6.5 minutes, which results in the temperature of the polypropylene increasing to 41°C. The current in the circuit is 3.2 A and the potential difference is 6 V. Find the specific heat capacity of polypropylene. Give your answer to 3 significant figures.
b) Why do we use an insulated container with a lid?
c) Mass appears in the specific heat capacity formula. How can we measure the mass of the liquid?
d) What piece of apparatus do we use to measure the change in temperature of the liquid?
3) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the unknown liquid was 300 grams, the change in temperature was 25°C and the reading on the joulemeter for change in energy was 9,750 joules. Work out the specific heat capacity of this liquid.
4) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the liquid is 150 grams. Before the immersion heater was switched on, the temperature of the liquid was 21°C.
The student leaves the immersion heater on until it transfers 15 kilojoules of energy to the liquid. After the immersion heater has transferred 15 kilojoules of energy to the liquid, the temperature of the liquid is 63°C.
Find the specific heat capacity of the liquid. Give your answer to 2 significant figures.
5) A student uses the apparatus in question 2 to find the specific heat capacity of an unknown liquid. The mass of the liquid is 350 grams. The student increases the temperature of the liquid by 20°C. She completes her experiment 3 times and the readings on the joulemeter are 13,032 J, 12,856 J and 12,962 J.
a) Find the mean energy transfer from the 3 experiments.
b) Find out the specific heat capacity of this liquid.
6) A student undertakes an experiment to find the specific heat capacity of marble. They use all of the apparatus in question 2 except a joulemeter. They put 300 grams of marble into the insulated container and turn the immersion heater on for 6 minutes. During these 6 minutes, the temperature of the marble increases by 55°C. The current in the circuit is 4.5 A and the potential difference is 9 V.
a) Find the power of the immersion heater.
b) Find the energy transferred by the immersion heater.
c) Find the specific heat capacity of marble. Give your answer to 2 significant figures.
7) A student undertakes an experiment to find the specific heat capacity of polypropylene. They use all of the apparatus in question 2 except a joulemeter. They put 170 grams of polypropylene into the insulated container. The initial temperature of the polypropylene is 18°C. The immersion heater is on for 6.5 minutes, which results in the temperature of the polypropylene increasing to 41°C. The current in the circuit is 3.2 A and the potential difference is 6 V. Find the specific heat capacity of polypropylene. Give your answer to 3 significant figures.