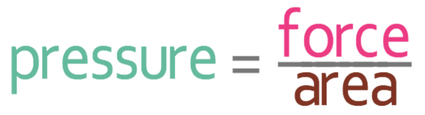Back to P3 Home
P3 B) Pressure in Gases
P3 B) Pressure in Gases
Pressure is a force per unit area. It is usually measured in N/m2, which means Newtons per square metre. We can work out pressure by dividing the force by the area. The calculation and formula triangle are shown below.
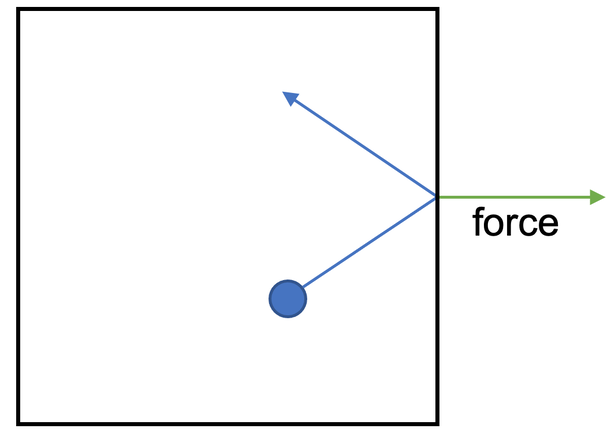
The particles in a gas move around in random directions and at different speeds. When the gas particles collide with other particles or the sides of the container, they exert a force. The diagram below shows the force exerted when a gas particle hits the side of the container.
When the gas particles hit the sides of the container, they exert a force at a right angle to the side of the container; this is a rightwards force on the above diagram.
If we were to increase the number of gas particles in the container, there would be more gas particles in the same volume. This will result in there being more frequent collisions between the gas particles and the sides of the container. This means that there will be a greater force exerted by the gas particles on the same area, thus resulting in a greater pressure (pressure is force divided by area). So, an increase in the number of gas particles results in pressure increasing.
If we were to decrease the volume of a container and keep the number of gas particles the same, there would be more gas particles in a given volume. This will mean that there will be more frequent collisions between the gas particles and the sides of the container, which will result in a greater force. Also, a decrease in the volume of a container will likely results in the area of the container becoming smaller. Pressure is force divided by area. So, a decrease in the volume of a container has resulted in a greater force and a smaller area, which will result in an increase in pressure. So, decreasing the volume of a container increases the pressure.
If we were to increase the number of gas particles in the container, there would be more gas particles in the same volume. This will result in there being more frequent collisions between the gas particles and the sides of the container. This means that there will be a greater force exerted by the gas particles on the same area, thus resulting in a greater pressure (pressure is force divided by area). So, an increase in the number of gas particles results in pressure increasing.
If we were to decrease the volume of a container and keep the number of gas particles the same, there would be more gas particles in a given volume. This will mean that there will be more frequent collisions between the gas particles and the sides of the container, which will result in a greater force. Also, a decrease in the volume of a container will likely results in the area of the container becoming smaller. Pressure is force divided by area. So, a decrease in the volume of a container has resulted in a greater force and a smaller area, which will result in an increase in pressure. So, decreasing the volume of a container increases the pressure.
An Increase in Temperature
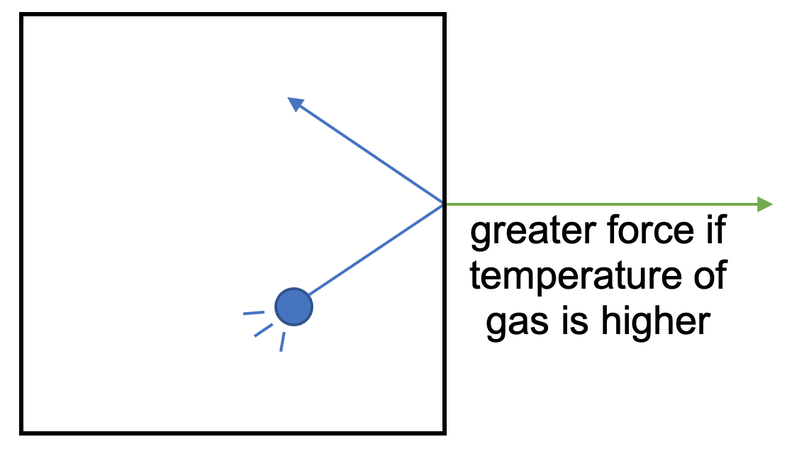
When we increase the temperature of a gas, we transfer energy to the kinetic energy stores of the gas particles, which results in the gas particles travelling around the container at a greater speed (the formula that connects kinetic energy and speed is ke = ½ mv2. An increase in kinetic energy (ke) of the particles means that the gas particles have a greater speed (v) – they move around faster).
An increase in temperature of a gas will result in pressure increasing (providing that the volume/ area of the container stays the same). This happens for 2 reasons:
- The gas particles will be moving around the container faster, which means that there will be more frequent collisions between the gas particles and the sides of the container.
- The gas particles will be moving around faster, which means that they have a greater momentum (momentum = mass x velocity; a greater velocity means a greater momentum). A greater momentum for the gas particles means that when the particles collide with the sides of the container, they exert a greater force.
So, when the temperature of a gas increases, there are more frequent collisions between the gas particles and the sides of the container, and a greater force exerted for each collision between a gas particle and the sides of the container. Both of these factors increase the total force on the sides of the container, which increases the pressure (providing that the area stays the same).