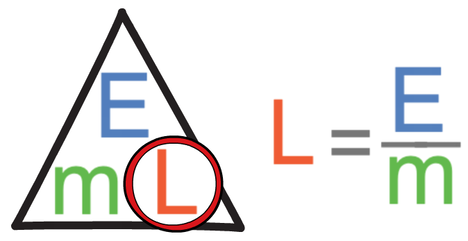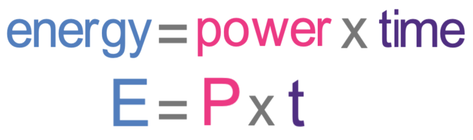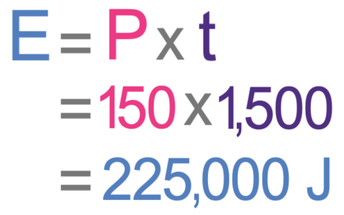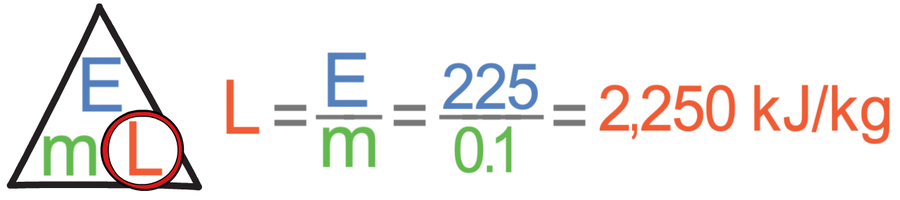Back to P3 Home
P3 G) Investigating Specific Latent Heat
P3 G) Investigating Specific Latent Heat
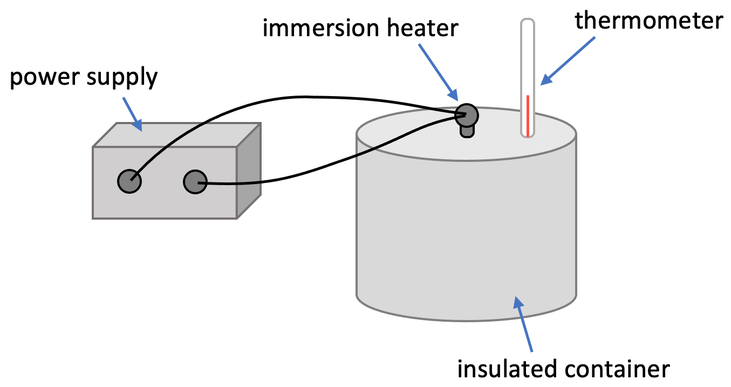
We can investigate the specific latent heat of fusion or vaporising by heating a certain mass of a substance with an immersion heater with a known power. Let’s suppose that our substance was 100 g of water and the power of the immersion heater is 150 watts. We set the apparatus up like what is shown below.
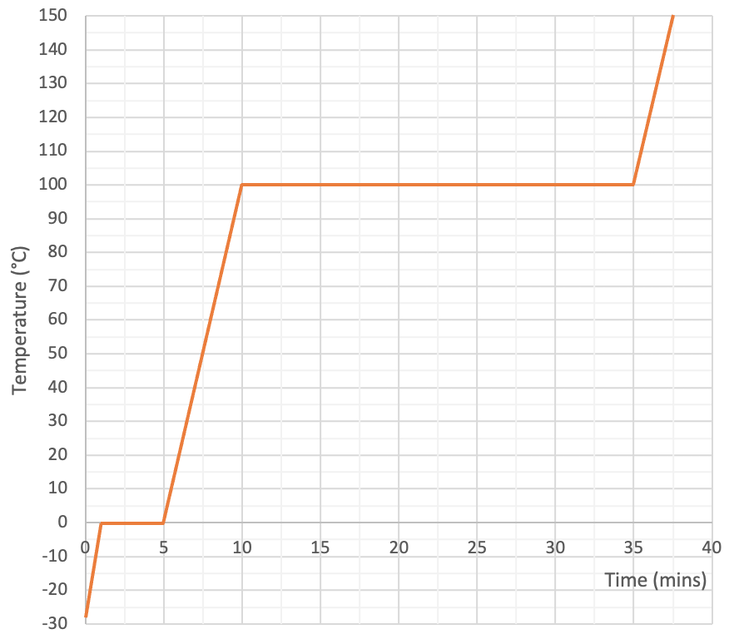
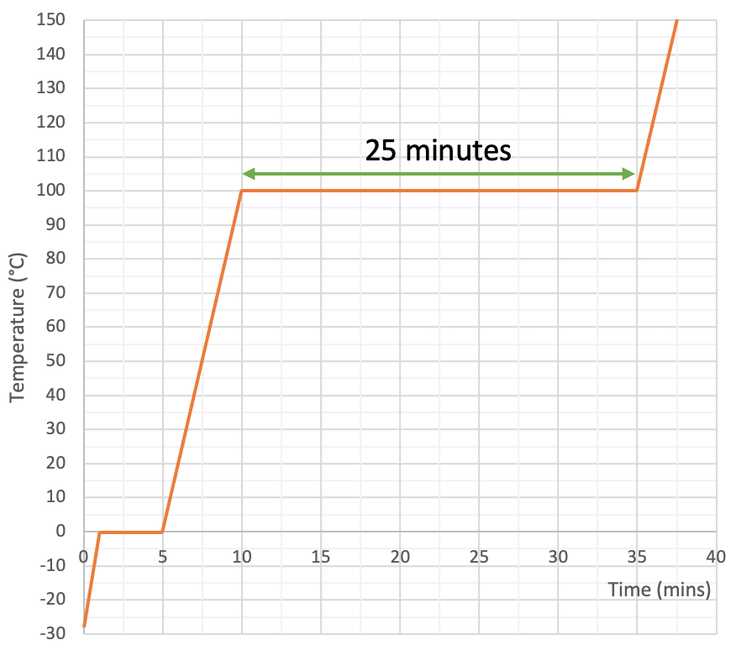
We then measure the temperature of the substance every minute and draw a heat curve like what is shown below.
The flat parts on the heat curve is where the substance is changing state. The flat part on the left is where the water changes from a solid to a liquid (melting). And the flat part on the right is where the water changes from a liquid to a gas (evaporating or boiling).
We can now use the values for the above graph to work out the specific latent heat energy of either fusion or vaporising.
Specific Latent Heat of Vaporisation
I am going to work out the specific latent heat of vaporisation, and we use the second flat part on the heat curve to do this. We work out the specific latent heat of vaporisation by dividing the energy (in kJ) by the mass (in kg).
At the start of this example, we were told that the mass of the water was 100 g. We need to have mass in kilograms rather than grams, so we divide 100 g by 1000, which is 0.1; the mass is 0.1 kg.
We now need to find the amount of energy transferred to the water during the second flat part. We are able to do this by multiplying the power of the heater by the time (in seconds).
We now need to find the amount of energy transferred to the water during the second flat part. We are able to do this by multiplying the power of the heater by the time (in seconds).
The question told us that the immersion heater has a power of 150 watts. One watt is 1 joule of energy per second. Therefore, our 150 watt immersion heater will transfer 150 joules of energy to the water every second. We now need to find the amount of time in seconds that the immersion heater was transferring energy to change the state of water from a liquid to a gas. We find the time by looking for the length of time for the second flat part on the graph. My working for this is shown on the graph below.
From the heat graph, we can see that the second flat part is 25 minutes long. We need to have our time in seconds. There are 60 seconds in 1 minute, so we multiply 25 by 60 to get 1,500 seconds.
We now have everything that we need to work out the amount of energy transferred; we have the power as 150 watts (or joules per second) and the time as 1,500 seconds. We sub these values into the formula:
We now have everything that we need to work out the amount of energy transferred; we have the power as 150 watts (or joules per second) and the time as 1,500 seconds. We sub these values into the formula:
This tells us that the amount of energy transferred is 225,000 joules.
For our specific latent heat calculation, we need to have our energy in kilojoules. There are 1,000 joules in 1 kilojoule, so we need to divide 225,000 by 1,000, which gives us 225 kJ.
We now have everything that we need in the correct units; we have the energy as 225 kJ and the mass as 0.1 kg. We sub these values into the calculation:
For our specific latent heat calculation, we need to have our energy in kilojoules. There are 1,000 joules in 1 kilojoule, so we need to divide 225,000 by 1,000, which gives us 225 kJ.
We now have everything that we need in the correct units; we have the energy as 225 kJ and the mass as 0.1 kg. We sub these values into the calculation:
This tells us that from our experiment, the specific latent heat of vaporisation for water is 2,250 kJ/kg.
The actual specific latent heat of vaporisation for water is 2,260 kJ/kg, so our calculation from our experiment is not too far off.
The actual specific latent heat of vaporisation for water is 2,260 kJ/kg, so our calculation from our experiment is not too far off.