1)
2)
a) When a substance changes state, the substance is exactly the same substance just in a different state. This means that a change in state is a physical change and not a chemical change (a chemical change would be when we end up with a difference substance/ a reaction takes place)
b) The mass and the number of particles in a substance remains the same when the substance changes state
3) Kinetic energy stores and potential energy stores
4)
a) Kinetic energy stores
b) The temperature of the substance increases
c) The total internal energy of the substance increases
5)
a) The potential energy stores
b) The temperature of the substance remains the same
c) The total internal energy of a substance increases
2)
a) When a substance changes state, the substance is exactly the same substance just in a different state. This means that a change in state is a physical change and not a chemical change (a chemical change would be when we end up with a difference substance/ a reaction takes place)
b) The mass and the number of particles in a substance remains the same when the substance changes state
3) Kinetic energy stores and potential energy stores
4)
a) Kinetic energy stores
b) The temperature of the substance increases
c) The total internal energy of the substance increases
5)
a) The potential energy stores
b) The temperature of the substance remains the same
c) The total internal energy of a substance increases
Questions
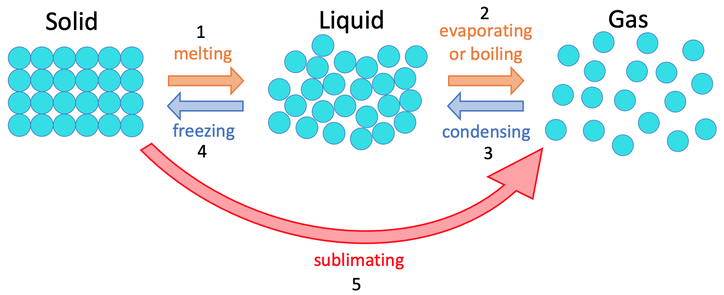
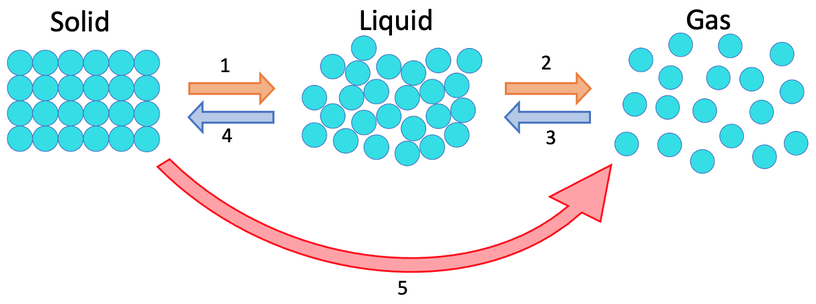
1) The three states of matter are shown below. Give the names for the 5 different changes of states.
1) The three states of matter are shown below. Give the names for the 5 different changes of states.
2)
a) Is a change in state a physical or chemical change?
b) Does the mass and the number of particles change when we change the state of a substance?
3) The total internal energy of a substance is the total energy that the particles have in 2 different stores; what are these two stores?
4)
a) When a substance is heated within a state, energy is transferred to which energy store?
b) What effect does this have on the temperature of the substance?
c) What effect does this have on the total internal energy of the substance?
5)
a) When a substance is heated at its melting or boiling point, energy is transferred to which energy store?
b) What effect does this have on the temperature of the substance?
c) What effect does this have on the total internal energy of the substance?
a) Is a change in state a physical or chemical change?
b) Does the mass and the number of particles change when we change the state of a substance?
3) The total internal energy of a substance is the total energy that the particles have in 2 different stores; what are these two stores?
4)
a) When a substance is heated within a state, energy is transferred to which energy store?
b) What effect does this have on the temperature of the substance?
c) What effect does this have on the total internal energy of the substance?
5)
a) When a substance is heated at its melting or boiling point, energy is transferred to which energy store?
b) What effect does this have on the temperature of the substance?
c) What effect does this have on the total internal energy of the substance?