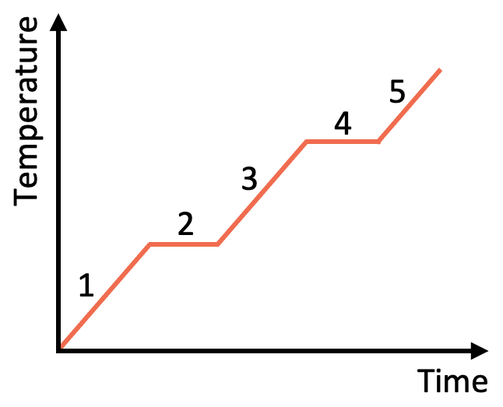
1)
1 = solid
2 = melting
3 = liquid
4 = boiling
5 = gas
b)
i) The energy is transferred to the kinetic energy stores of the particles in the substance, which result in the temperature of the substance increasing
ii) The energy goes to breaking the intermolecular forces attraction between the particles (the energy goes to increasing the potential energy stores of the particles). The temperature of the substance does not increase
2)
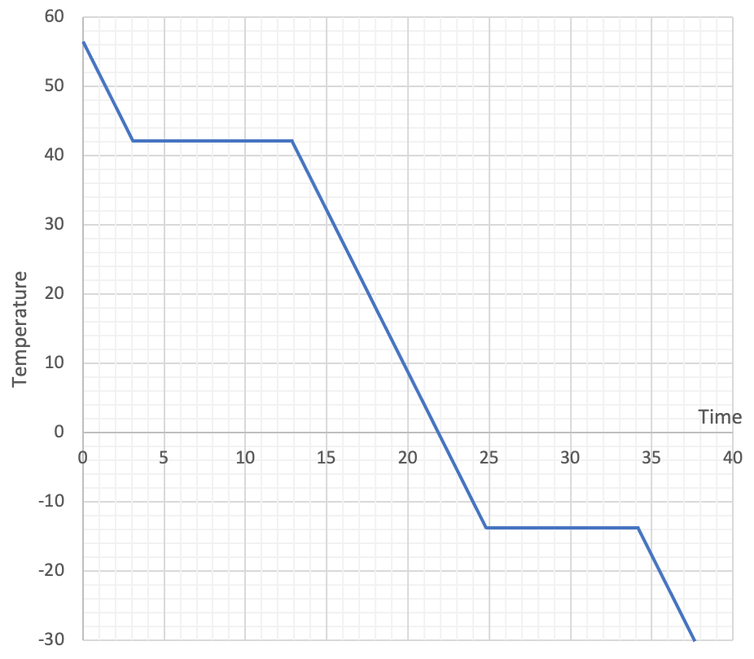
a) 42°C (±1°C)
b)
i) The internal energy of a substance decreases
ii) During a change down a state (gas to liquid, or liquid to solid), intermolecular bonds are formed. The forming of these intermolecular bonds releases energy, which is why the temperature of the substance remains the same when it goes down a change in state
c) -14°C (±1°C)
d) When we cool a substance within a state, energy is lost from the particle’s kinetic energy stores. This results in the temperature of the substance decreasing
3)
a) Specific latent heat is the amount of energy required to change the state of 1 kg of a substance without changing the temperature of the substance
b) Melting (solid to liquid) or freezing (liquid to solid)
c) Evaporation (liquid to gas) or condensation (gas to liquid)
d) Energy = mass x SLH
E = mL
1 = solid
2 = melting
3 = liquid
4 = boiling
5 = gas
b)
i) The energy is transferred to the kinetic energy stores of the particles in the substance, which result in the temperature of the substance increasing
ii) The energy goes to breaking the intermolecular forces attraction between the particles (the energy goes to increasing the potential energy stores of the particles). The temperature of the substance does not increase
2)
a) 42°C (±1°C)
b)
i) The internal energy of a substance decreases
ii) During a change down a state (gas to liquid, or liquid to solid), intermolecular bonds are formed. The forming of these intermolecular bonds releases energy, which is why the temperature of the substance remains the same when it goes down a change in state
c) -14°C (±1°C)
d) When we cool a substance within a state, energy is lost from the particle’s kinetic energy stores. This results in the temperature of the substance decreasing
3)
a) Specific latent heat is the amount of energy required to change the state of 1 kg of a substance without changing the temperature of the substance
b) Melting (solid to liquid) or freezing (liquid to solid)
c) Evaporation (liquid to gas) or condensation (gas to liquid)
d) Energy = mass x SLH
E = mL
4) 1,254 kJ
5) 904 kJ
6) 54,990 J
Questions
1) The graph below is a heating graph for the heating of a substance.
1) The graph below is a heating graph for the heating of a substance.
a) Name all of the 5 labels on the above heating graph. Use the words melting, boiling, solid, liquid and gas.
b) As time progresses, more energy is transferred to the substance.
i) When we stay within a state, what energy store is this energy transferred to? Does the temperature of the substance increase?
ii) When we are at the melting or boiling point, what does the energy transferred to the substance do? Does the temperature of the substance increase?
2) The graph below is the heating graph for the cooling of a substance.
b) As time progresses, more energy is transferred to the substance.
i) When we stay within a state, what energy store is this energy transferred to? Does the temperature of the substance increase?
ii) When we are at the melting or boiling point, what does the energy transferred to the substance do? Does the temperature of the substance increase?
2) The graph below is the heating graph for the cooling of a substance.
a) What is the temperature of the boiling point?
b) When a substance is going down a change in state (gas to liquid or liquid to solid);
i) does the internal energy of the substance increase or decrease?
ii) Why does the temperature not change?
c) What is the temperature of the melting point?
d) When we remain within a state when we are cooling a substance, energy is lost from the internal energy stores of the substance. What internal energy store loses energy? Does this result in the temperature of the substance increasing, decreasing or staying the same?
3)
a) Define specific latent heat.
b) What state change is the specific latent heat of fusion for? Do not worry about the direction.
c) What state change is the specific latent heat of vaporisation for? Do not worry about the direction.
d) Write down the formula that links specific latent heat, mass and energy. Also, write down the formula triangle.
4) 6 kilograms of iron is already at its melting point. The specific latent heat of fusion for iron is 209 kJ/kg. How much energy is required to change 6 kg of iron from a solid to a liquid at its melting point? Give your answer in kilojoules.
5) How much energy is needed to change 400 grams of water from a liquid to a gas at 100°C? The specific latent heat of vaporisation for water is 2,260 kJ/kg. Give your answer in kilojoules.
6) The specific latent heat of vaporisation for ethanol is 846 kJ/kg. We have 65 grams of ethanol that is already at its boiling point. Work out the amount of energy required to turn 65 grams of water from a liquid to a gas at its boiling point. Give your answer in joules.
b) When a substance is going down a change in state (gas to liquid or liquid to solid);
i) does the internal energy of the substance increase or decrease?
ii) Why does the temperature not change?
c) What is the temperature of the melting point?
d) When we remain within a state when we are cooling a substance, energy is lost from the internal energy stores of the substance. What internal energy store loses energy? Does this result in the temperature of the substance increasing, decreasing or staying the same?
3)
a) Define specific latent heat.
b) What state change is the specific latent heat of fusion for? Do not worry about the direction.
c) What state change is the specific latent heat of vaporisation for? Do not worry about the direction.
d) Write down the formula that links specific latent heat, mass and energy. Also, write down the formula triangle.
4) 6 kilograms of iron is already at its melting point. The specific latent heat of fusion for iron is 209 kJ/kg. How much energy is required to change 6 kg of iron from a solid to a liquid at its melting point? Give your answer in kilojoules.
5) How much energy is needed to change 400 grams of water from a liquid to a gas at 100°C? The specific latent heat of vaporisation for water is 2,260 kJ/kg. Give your answer in kilojoules.
6) The specific latent heat of vaporisation for ethanol is 846 kJ/kg. We have 65 grams of ethanol that is already at its boiling point. Work out the amount of energy required to turn 65 grams of water from a liquid to a gas at its boiling point. Give your answer in joules.