1)
a) Specific latent heat is the amount of energy required to change the state of 1 kg of a substance without changing the temperature of the substance
b) Energy = mass x SLH
E = mL
a) Specific latent heat is the amount of energy required to change the state of 1 kg of a substance without changing the temperature of the substance
b) Energy = mass x SLH
E = mL
2)
a) 7 minutes
b) 50,400 J or 50.4 KJ
c) 336 kJ/kg
Questions
1)
a) Define specific latent heat.
b) Write down the formula that links specific latent heat, mass and energy. Also, write down the formula triangle.
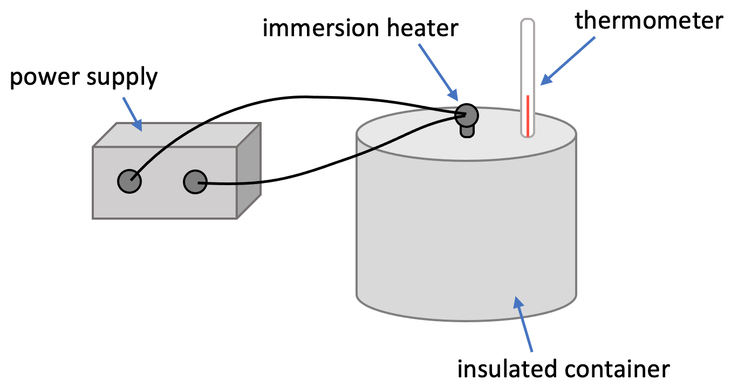
2) A student completed an experiment to find the specific latent heat of fusion for water. She put 150 grams of ice (water) into an insulated beaker and used a 120 watt immersion heater to heat the water. She sets up the apparatus like what is shown below.
1)
a) Define specific latent heat.
b) Write down the formula that links specific latent heat, mass and energy. Also, write down the formula triangle.
2) A student completed an experiment to find the specific latent heat of fusion for water. She put 150 grams of ice (water) into an insulated beaker and used a 120 watt immersion heater to heat the water. She sets up the apparatus like what is shown below.
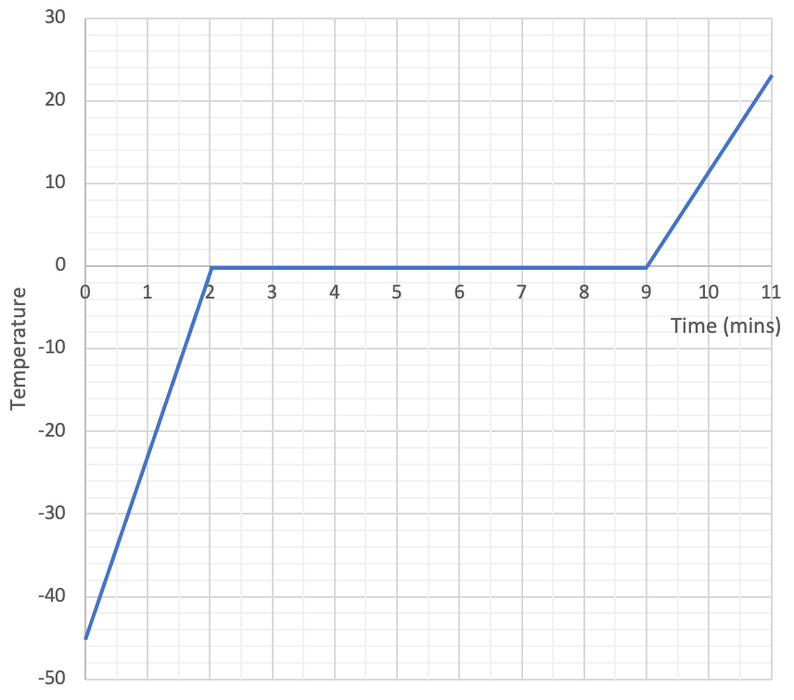
She then measures the temperature every 30 seconds and draws the heating graph below.
a) The only flat part on the graph is where the ice changes from a solid to a liquid. How long is this flat part?
b) How much energy is transferred during this flat part?
c) Work out the specific latent heat of fusion for water. Give your answer in kJ/kg
b) How much energy is transferred during this flat part?
c) Work out the specific latent heat of fusion for water. Give your answer in kJ/kg