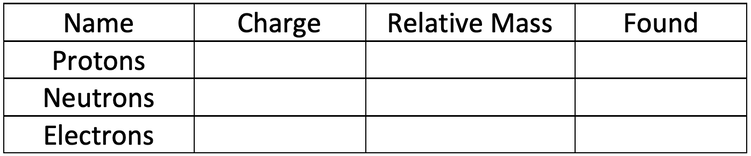
1) The current model of the atom that we use is the nuclear model. Fill in the table below for the charge, mass and found (location within the atom) for the three different subatomic particles.
2)
a) What is the charge of elements; positive, negative or neutral?
b) Are the number of protons and electrons in an element the same or different?
c) The tile for fluorine in the periodic table is shown below.
Write down the number of protons, neutrons and electrons for fluorine.
3)
a) What is an ion?
b) If an atom has more protons than electrons, are they a positive or negative ion?
c) If an atom has more electrons than protons, are they a positive or negative ion?
4) What is an isotope?
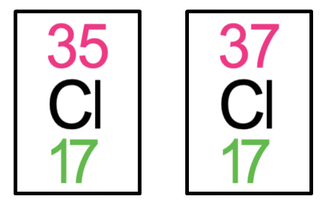
5) The tiles for chlorine-35 and chlorine-37 are both shown below.
3)
a) What is an ion?
b) If an atom has more protons than electrons, are they a positive or negative ion?
c) If an atom has more electrons than protons, are they a positive or negative ion?
4) What is an isotope?
5) The tiles for chlorine-35 and chlorine-37 are both shown below.
a) Find the number of protons and neutrons for both chlorine-35 and chlorine-37.
b) What is a similarity between chlorine-35 and chlorine-37?
c) What is a difference between chlorine-35 and chlorine-37?
b) What is a similarity between chlorine-35 and chlorine-37?
c) What is a difference between chlorine-35 and chlorine-37?