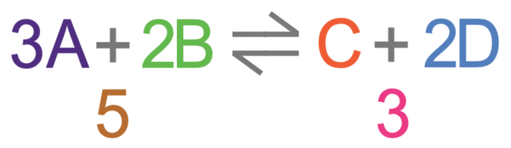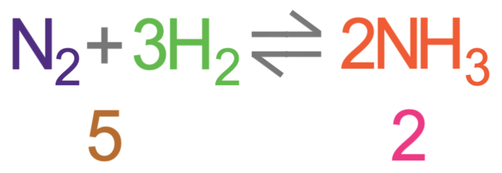C4 I) Dynamic Equilibriums
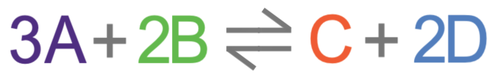
We can talk about the positions of dynamic equilibriums by saying which side the equilibrium lies. If we say that the equilibrium lies to the right, it means that there is a greater concentration of the products (C and D) compared to the reactants (A and B). If we say that the equilibrium lies to the left, it means that there is a greater concentration of the reactants (A and B) compared to the products (C and D).
The position of the dynamic equilibrium is dependent upon the conditions that the reversible reaction takes place in. The position of the dynamic equilibrium is dependent upon temperature, pressure (only for gases) and concentration. Le Chatelier’s principles states that if we change any of these factors (temperature, pressure or concentration), the equilibrium position would move to counteract the change. For example, if we increased the temperature, the equilibrium would move to decrease the temperature. If we decrease the pressure, the equilibrium would move to increase the pressure.
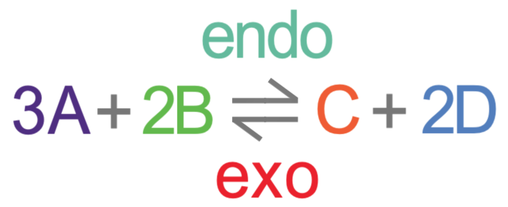
The affect that temperature has on the dynamic equilibrium depends on the type of reaction. Reversible reactions go in both directions, and one of the directions will be endothermic and the other direction will be exothermic.
Le Chatelier’s principles states that the reversible reaction will try to counteract the change that we make.
For example, if we increase the temperature that the reaction takes place in, the reversible reaction will want to counteract the change by taking in/ removing this extra heat to reverse the temperature increase. This will mean that more of the endothermic reactions will take place as endothermic reactions take in heat/ energy. Therefore, increasing the temperature favours the endothermic reaction. For the reversible reaction at the top, the forwards reaction was endothermic, and this means that an increase in temperature will result in more of the forwards reactions taking place, which will move the position of the equilibrium towards the right; we will have an equilibrium with more C and D, and less A and B.
Here is a summary of the effects that temperature has:
- An increase in temperature favours the endothermic reaction as the reversible reaction will want to take in the extra heat/ energy. This means that the equilibrium moves towards the endothermic side.
- A decrease in temperature favours the exothermic reaction as the reversible reaction will want to release/ produce more heat/ energy. This means that the equilibrium moves towards the exothermic side.
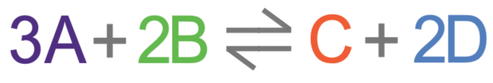
Pressure is only relevant for gases. Pressure is a force per unit of area. We can increase or decrease the pressure in the closed system that the reversible reaction is taking place in. In order to know what the effect is for pressure, we need to count the molecules that are on each side of the reversible reaction. Our made-up reversible reaction is shown below.
If we decrease pressure, the dynamic equilibrium will move towards the side of the reaction that has more molecules. This is because more molecules will increase the pressure, thus reversing the pressure decrease. For our reaction, a decrease in pressure will result in the equilibrium moving towards the left as there are more molecules of A and B (5), and less of C and D (3). Therefore, a decrease in pressure results in an equilibrium with more A and B, and less C and D.
Here is a summary of the effects that pressure has:
- An increase in pressure favours the side with fewer gas molecules. This is because fewer gas molecules will decrease the pressure reversing the pressure increase.
- A decrease in pressure favours the side with more gas molecules. This is because more gas molecules will increase the pressure reversing the pressure decrease.
In the above passage, I have spoken about the number of molecules of gas. Instead of saying molecules of gas, you can say moles of gas. So, for the above reaction, the left side has 5 moles of gas and the right side has 3 moles of gas.
Concentration refers to the mass of the reactants or products. If we change the concentration of any of the substances, the reversible reaction will respond to counteract the change that we have made.
- If we increase the quantity of reactants (A and B), the dynamic equilibrium will move towards the right to counteract the increase in A and B. This is because after the increase in A and B, there is now more of the reactants, which means that more forward reactions will take place resulting in more C and D, and less A and B. The equilibrium moves to the right.
- If we decreased the reactants (A and B), the equilibrium would move towards the left. This is because after the decrease in A and B, there is now more C and D relative to A and B. This means that more backward reactions will occur compared to the forward reactions, which results in the equilibrium moving towards the left; there will be more A and B, and less C and D.
- If we increased the quantity of the products (C and D), the dynamic equilibrium will move to the left because C and D will react to produce A and B.
- If we decrease the quantity of the products (C and D), the dynamic equilibrium will move to the right, because A and B will react together to produce more of C and D.
We are now going to have a look at the Haber process, which is a reversible reaction. The reaction is shown below.
From looking at the equation above, we can see that the forwards reaction produces heat. This means that the forwards reaction is exothermic, and the backwards reaction is endothermic.
The aim of the Haber process is to produce ammonia (NH3). We are now going to look at the conditions that will give us a dynamic equilibrium with as much ammonia as possible; the conditions that will move the dynamic equilibrium to the right. The conditions that we will look at will be for temperature and pressure.
For temperature, the forwards reaction is exothermic. This means that if we reduce the temperature, more of the exothermic reactions will take place to increase temperature, thus meaning that more forwards reactions take place and more ammonia is produced. Therefore, we want a low temperature.
In order to work out the pressure that gives us the most ammonia, we need to count the molecules on either side of the reaction. On the left side of the reaction, there are 4 molecules of gas (1 from N2 and 3 from H2), and on the right side there are 2 molecules of gas (from the NH3). Therefore, in order to obtain an equilibrium with as much ammonia as possible, we will want to have a high pressure because a high pressure will favour the side with fewer gas molecules, thus meaning that there will be more ammonia (the equilibrium moves towards the right).
So, the conditions where the dynamic equilibrium will have the most ammonia will be at a low temperature and a high pressure.
However, these are not the conditions that we produce ammonia at in industry because we need to think about the cost implications of such conditions.