Catalyst have two characteristics; they increase the rate of reaction and are not used up during the reaction. This means that after a reaction has taken place, the catalyst is free to catalyse other reactions. This is why we only need a small amount of a catalyst to catalyse a large mass of reactants. Catalysts only catalyse certain reactions. An example of a catalyst is vanadium (V) oxide, which is the catalyst that is used in the production of sulphuric acid. Another example of a catalyst is iron in the Haber process (the Haber process is the reaction of nitrogen and hydrogen to produce ammonia).
Catalysts increase the rate of reactions by offering an alternative reaction pathway for the reaction that requires a lower activation energy. Activation energy is the amount of energy that is required to start a reaction. If the activation energy is not met, the reaction will not take place. If the activation energy is met, the reaction will take place.
Catalysts increase the rate of reactions by offering an alternative reaction pathway for the reaction that requires a lower activation energy. Activation energy is the amount of energy that is required to start a reaction. If the activation energy is not met, the reaction will not take place. If the activation energy is met, the reaction will take place.
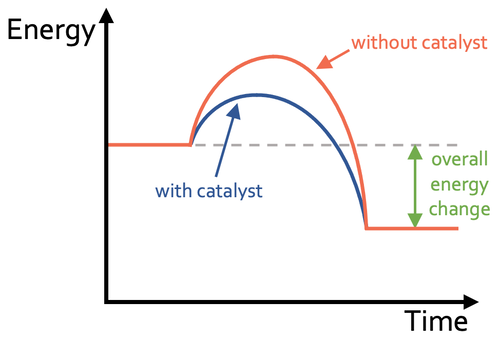
The reaction profile above shows the reaction pathway when a catalyst is used (blue) and when a catalyst is not used (orange). The activation energy is the difference between the energy in the reactants and the highest energy point on the reaction profile. We can see from the above graph that a lower activation energy is required when the catalyst is present.
We can also see from the graph that the catalyst does not alter the amount of energy in the reactants or products; the overall energy change during the reaction is the same (the overall energy change is the difference between the energy in the products and the energy in the reactants). The catalyst just offers an alternative pathway during the reaction that requires a lower activation energy.
Catalysts are useful in industry because a lower activation energy is required when a catalyst is present. This means that reactions with catalysts can take place at lower temperatures, which requires less energy, thus reducing the costs for the manufacturing process (the energy/ temperature in reactions in industry comes from burning fuels, so a lower temperature means less fuel is used and therefore lower costs, which means more profit for the firm).
We can also see from the graph that the catalyst does not alter the amount of energy in the reactants or products; the overall energy change during the reaction is the same (the overall energy change is the difference between the energy in the products and the energy in the reactants). The catalyst just offers an alternative pathway during the reaction that requires a lower activation energy.
Catalysts are useful in industry because a lower activation energy is required when a catalyst is present. This means that reactions with catalysts can take place at lower temperatures, which requires less energy, thus reducing the costs for the manufacturing process (the energy/ temperature in reactions in industry comes from burning fuels, so a lower temperature means less fuel is used and therefore lower costs, which means more profit for the firm).
Catalyst Investigation
In order for something to be a catalyst, it must increase the rate of reaction AND not be used up during the reaction.
If you are asked to test whether a solid is a catalyst for a particular reaction between two solutions, you must first make sure that the solid that you think may be a catalyst remains unchanged during the reaction. If the potential catalyst does remain unchanged, it tells us that the solid was not used in the reaction and therefore it may be a catalyst. We check whether the potential solid catalyst has remained unchanged during the reaction by measuring the mass of the potential catalyst before and after the reaction. If the masses before and after the reaction are the same, it tells us that the potential solid catalyst has not been used up and therefore may be a catalyst (providing that it also increases the rate of reaction). If the masses are not the same, it tells us that the solid catalyst has reacted and is therefore not a catalyst. We are able to find the mass of the potential catalyst after the reaction has taken place by filtering the solution, which will give us our solid (remember, we use filtration to separate an insoluble solid from a solution).
The next step is to check whether the potential catalyst increases the rate of reaction. We do this by carrying out two different reactions; one reaction will be with the potential catalyst, and the other reaction will be without the potential catalyst. Both of these reactions should be carried out with everything else the same, such as the same temperature, the same quantity of reactants etc…. We then compare the rates of reaction for the two reactions. If the rate of reaction increases with the potential catalyst, then we can say that the solid is a catalyst. If the rate of reaction stays the same, then we can say that the solid is not a catalyst.
So, to prove that a solid is a catalyst, you need to prove that it increases the rate of reaction AND is not used up during the reaction.
In order for something to be a catalyst, it must increase the rate of reaction AND not be used up during the reaction.
If you are asked to test whether a solid is a catalyst for a particular reaction between two solutions, you must first make sure that the solid that you think may be a catalyst remains unchanged during the reaction. If the potential catalyst does remain unchanged, it tells us that the solid was not used in the reaction and therefore it may be a catalyst. We check whether the potential solid catalyst has remained unchanged during the reaction by measuring the mass of the potential catalyst before and after the reaction. If the masses before and after the reaction are the same, it tells us that the potential solid catalyst has not been used up and therefore may be a catalyst (providing that it also increases the rate of reaction). If the masses are not the same, it tells us that the solid catalyst has reacted and is therefore not a catalyst. We are able to find the mass of the potential catalyst after the reaction has taken place by filtering the solution, which will give us our solid (remember, we use filtration to separate an insoluble solid from a solution).
The next step is to check whether the potential catalyst increases the rate of reaction. We do this by carrying out two different reactions; one reaction will be with the potential catalyst, and the other reaction will be without the potential catalyst. Both of these reactions should be carried out with everything else the same, such as the same temperature, the same quantity of reactants etc…. We then compare the rates of reaction for the two reactions. If the rate of reaction increases with the potential catalyst, then we can say that the solid is a catalyst. If the rate of reaction stays the same, then we can say that the solid is not a catalyst.
So, to prove that a solid is a catalyst, you need to prove that it increases the rate of reaction AND is not used up during the reaction.