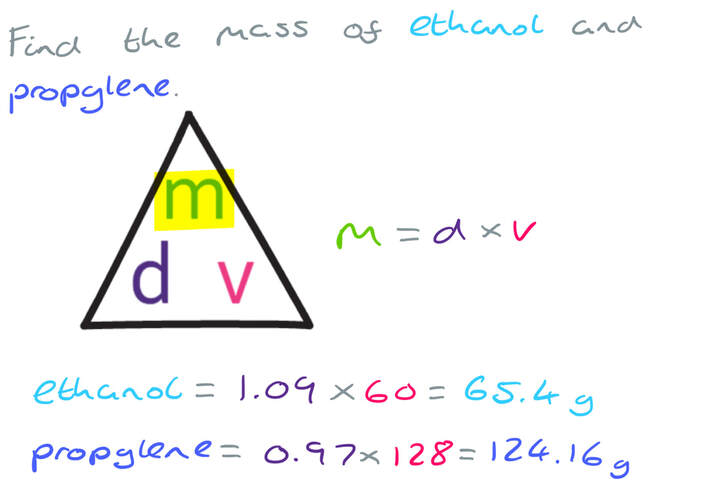Back to Edexcel Maths Higher June 2019 Paper 3 Home
Q13: Answers – Paper 3 – June 2019
Q13: Answers – Paper 3 – June 2019
Helpful Links
Mark Scheme
Working
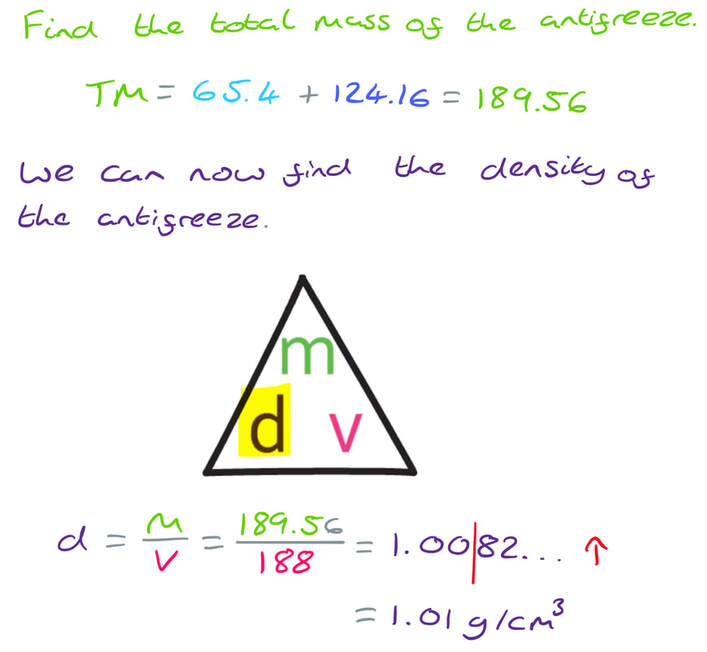
The density of ethanol is 1.09 g/cm3
The density of propylene is 0.97 g/cm3
60 litres of ethanol are mixed with 128 litres of propylene to make 188 litres of antifreeze.
Work out the density of the antifreeze.
Give your answer correct to 2 decimal places.
Question
13)
The density of ethanol is 1.09 g/cm3
The density of propylene is 0.97 g/cm3
60 litres of ethanol are mixed with 128 litres of propylene to make 188 litres of antifreeze.
Work out the density of the antifreeze.
Give your answer correct to 2 decimal places.
[4 marks]