Back to C1 Home
C1: Quiz 8 – Answers
C1: Quiz 8 – Answers
1)
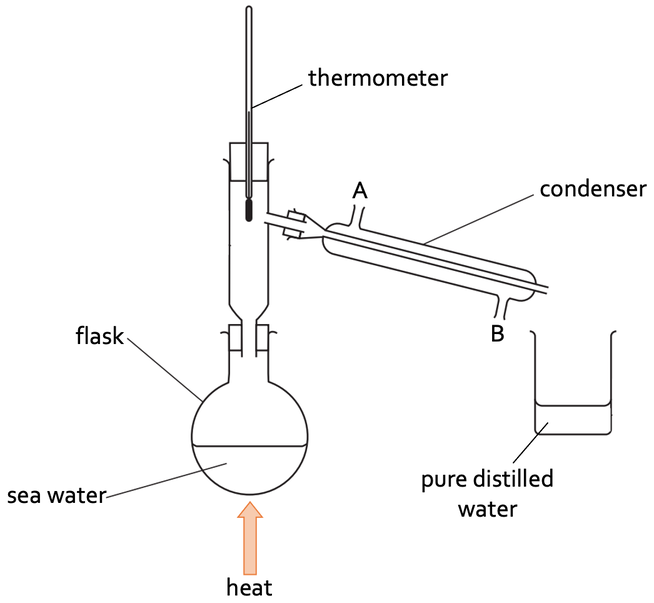
a) We apply heat to the sea water (usually with a Bunsen burner). This will result in the water evaporating (turning from a liquid to a gas) and rising up the flask. The water vapor then moves down the condenser where it condenses (turns from a gas to a liquid). The liquid water then runs into the collecting beaker at the end of the condenser. The salt will remain in the flask that was being heated. We also have a thermometer on the top of the flask to ensure that the flask is the correct temperature for the boiling point of the liquid in the solution.
b)
i) Cold water enters the condenser through hole B
ii) Cold water leaves the condenser through hole A
2) We would use fractional distillation when we have a mixture of liquids with similar boiling points
3)
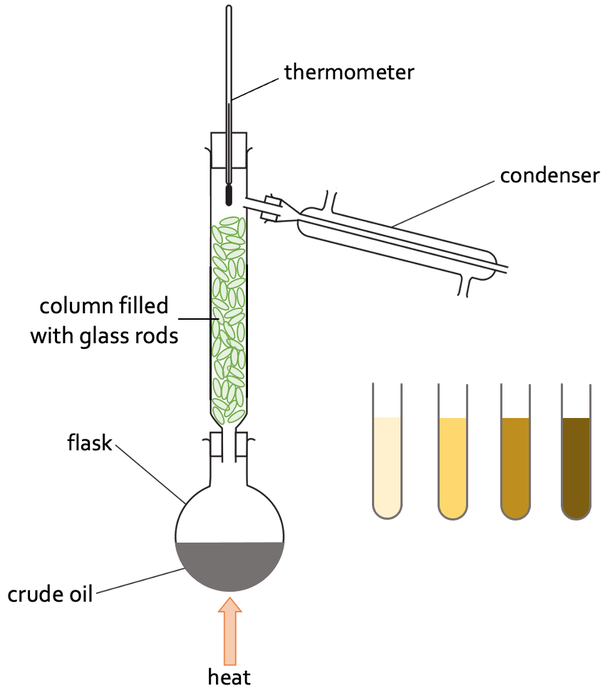
a) We always start with the lowest temperature and then slowly increase the temperature. This is because if we went from the highest temperature to the lowest temperature, the whole mixture of liquids would evaporate, rise up the column, down the condenser and run into the collection beaker. This would result in the collection beaker containing a mixture of liquids.
b)
i) The column is the hottest at the bottom
ii) The column is the coolest at the top
c) Other liquids in the mixture with higher boiling points may also start to evaporate and rise up the column filled with glass rods. The top of the column is cooler than the bottom of the column, which results in the vapours of the fractions with higher boiling points condensing part way up the column and running back down the glass rods in the column to the flask. The column filled with glass rods ensures that only the fraction with the lowest boiling point (the fraction that we want) will rise up the column, down the condenser and into the collection beaker; the column with glass rods makes sure that we do not get any unwanted fractions in the collection beaker on the other side of the condenser.
4)
a)
i) Propanol as it has the lowest boiling point
ii) Butanol as it has the next lowest boiling point/ the middle boiling point
iii) Pentanol as it has the highest boiling point
b) The student will use the thermometer at the top of the column to ensure that the temperature is the correct temperature for the boiling points of the fractions
a) We apply heat to the sea water (usually with a Bunsen burner). This will result in the water evaporating (turning from a liquid to a gas) and rising up the flask. The water vapor then moves down the condenser where it condenses (turns from a gas to a liquid). The liquid water then runs into the collecting beaker at the end of the condenser. The salt will remain in the flask that was being heated. We also have a thermometer on the top of the flask to ensure that the flask is the correct temperature for the boiling point of the liquid in the solution.
b)
i) Cold water enters the condenser through hole B
ii) Cold water leaves the condenser through hole A
2) We would use fractional distillation when we have a mixture of liquids with similar boiling points
3)
a) We always start with the lowest temperature and then slowly increase the temperature. This is because if we went from the highest temperature to the lowest temperature, the whole mixture of liquids would evaporate, rise up the column, down the condenser and run into the collection beaker. This would result in the collection beaker containing a mixture of liquids.
b)
i) The column is the hottest at the bottom
ii) The column is the coolest at the top
c) Other liquids in the mixture with higher boiling points may also start to evaporate and rise up the column filled with glass rods. The top of the column is cooler than the bottom of the column, which results in the vapours of the fractions with higher boiling points condensing part way up the column and running back down the glass rods in the column to the flask. The column filled with glass rods ensures that only the fraction with the lowest boiling point (the fraction that we want) will rise up the column, down the condenser and into the collection beaker; the column with glass rods makes sure that we do not get any unwanted fractions in the collection beaker on the other side of the condenser.
4)
a)
i) Propanol as it has the lowest boiling point
ii) Butanol as it has the next lowest boiling point/ the middle boiling point
iii) Pentanol as it has the highest boiling point
b) The student will use the thermometer at the top of the column to ensure that the temperature is the correct temperature for the boiling points of the fractions
Questions
1) The diagram below shows how we can separate sea water to obtain pure distilled water.
1) The diagram below shows how we can separate sea water to obtain pure distilled water.
a) Describe how we can use the apparatus above to obtain pure distilled water.
b) The condenser has two holes in it where cold water from a tap enters and leaves the condenser; these holes are labelled as A and B on the above diagram.
i) Through which hole does cold water enter the condenser?
ii) Through which hole does cold water leave the condenser?
2) When would we use fractional distillation rather than simple distillation?
3) The set up for fractional distillation of crude oil is shown below.
b) The condenser has two holes in it where cold water from a tap enters and leaves the condenser; these holes are labelled as A and B on the above diagram.
i) Through which hole does cold water enter the condenser?
ii) Through which hole does cold water leave the condenser?
2) When would we use fractional distillation rather than simple distillation?
3) The set up for fractional distillation of crude oil is shown below.
a) When completing fractional distillation, do we go from a low temperature to a high temperature, or from a high temperature to a low temperature? Explain your answer.
b)
i) Where is the column filled with glass rods the hottest?
ii) Where is the column filled with glass rods the coolest?
c) Describe the role that the long column filled with glass rods has on the process.
4) A student completes fractional distillation on a mixture of 3 liquids. The three liquids and their boiling points are:
a) When a student completes fractional distillation on the mixture containing these 3 liquids, which of the liquids will be:
i) the first fraction that she obtains? Explain your answer.
ii) the second fraction that she obtains? Explain your answer.
iii) the third fraction that she obtains? Explain your answer.
b) The student uses the same apparatus that is shown in question 3. What piece of equipment will she use to ensure that just one of the fractions will evaporate?
b)
i) Where is the column filled with glass rods the hottest?
ii) Where is the column filled with glass rods the coolest?
c) Describe the role that the long column filled with glass rods has on the process.
4) A student completes fractional distillation on a mixture of 3 liquids. The three liquids and their boiling points are:
- Butanol: 118°C
- Propanol: 97°C
- Pentanol: 138°C
a) When a student completes fractional distillation on the mixture containing these 3 liquids, which of the liquids will be:
i) the first fraction that she obtains? Explain your answer.
ii) the second fraction that she obtains? Explain your answer.
iii) the third fraction that she obtains? Explain your answer.
b) The student uses the same apparatus that is shown in question 3. What piece of equipment will she use to ensure that just one of the fractions will evaporate?