Back to C2 Home
C2: Quiz 5 – Answers
C2: Quiz 5 – Answers
1) Strong, weak
Simple molecular substances have extremely STRONG covalent bonds that join all of the atoms within a simple molecular substance together. The intermolecular forces of attraction for simple molecular substances are very WEAK.
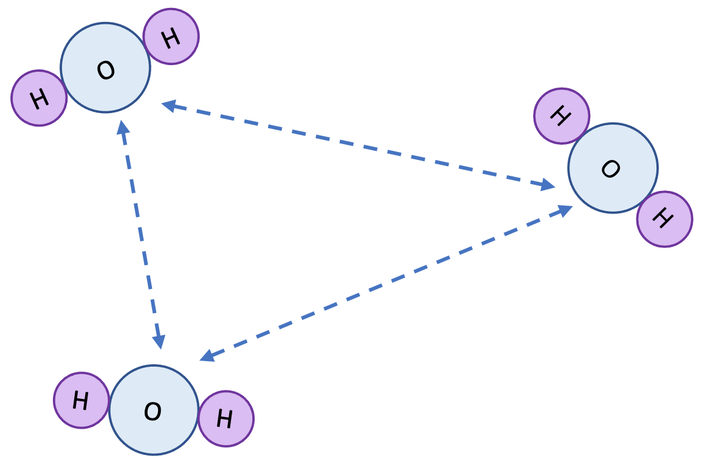
2) Intermolecular bonds/ intermolecular forces of attraction
3) Low melting and boiling points because they have weak intermolecular forces of attraction, which means that little energy is required to break/ overcome these weak intermolecular forces and change their states
4) Simple molecular substances do not conduct electricity as they do not have delocalised electrons or ions
5) Increase (as the simple molecular substances get larger, the melting and boiling points increase)
6)
a) Dodecane has the highest melting and boiling point because it is the largest molecule and therefore has the strongest intermolecular forces of attraction, which requires more energy to break, thus meaning that dodecane has the highest melting and boiling point
b) Methane has the lowest melting and boiling point because it is the smallest molecule and therefore has the weakest intermolecular forces of attraction, which requires little energy to break, thus meaning that methane has the lowest melting and boiling point
Simple molecular substances have extremely STRONG covalent bonds that join all of the atoms within a simple molecular substance together. The intermolecular forces of attraction for simple molecular substances are very WEAK.
2) Intermolecular bonds/ intermolecular forces of attraction
3) Low melting and boiling points because they have weak intermolecular forces of attraction, which means that little energy is required to break/ overcome these weak intermolecular forces and change their states
4) Simple molecular substances do not conduct electricity as they do not have delocalised electrons or ions
5) Increase (as the simple molecular substances get larger, the melting and boiling points increase)
6)
a) Dodecane has the highest melting and boiling point because it is the largest molecule and therefore has the strongest intermolecular forces of attraction, which requires more energy to break, thus meaning that dodecane has the highest melting and boiling point
b) Methane has the lowest melting and boiling point because it is the smallest molecule and therefore has the weakest intermolecular forces of attraction, which requires little energy to break, thus meaning that methane has the lowest melting and boiling point
Questions
1) Fill in the passage below using the words “strong” and “weak”.
Simple molecular substances have extremely ________ covalent bonds that join all of the atoms within a simple molecular substance together. The intermolecular forces of attraction for simple molecular substances are very ________.
2) What do the dashed lines on the diagram below represent?
1) Fill in the passage below using the words “strong” and “weak”.
Simple molecular substances have extremely ________ covalent bonds that join all of the atoms within a simple molecular substance together. The intermolecular forces of attraction for simple molecular substances are very ________.
2) What do the dashed lines on the diagram below represent?
3) Do simple molecular substances have high or low melting and boiling points? Explain your answer.
4) Do simple covalent substances conduct electricity? Explain your answer.
5) Do the melting and boiling points increase or decrease as the sizes of the simple molecular substances increase?
6) We have three hydrocarbons; methane (CH4), pentane (C5H12) and dodecane (C12H26).
a) Which substance has the highest melting and boiling point? Explain your answer.
b) Which substance has the lowest melting and boiling point? Explain your answer.