Back to C2 Home
C2: Quiz 6 – Answers
C2: Quiz 6 – Answers
1)
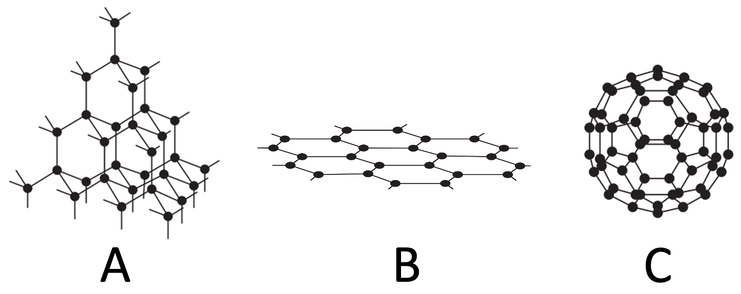
a) Diamond
b) Graphene
c) Buckminster/ fullerenes
2)
a) 4
b) Very high melting and boiling points because diamond is a giant covalent structure that has loads of covalent bonds. These take a lot of energy to break and change state, thus meaning that diamond has a very high melting and boiling point
c) Diamond does not conduct electricity as there are no delocalised electrons or ions that can move
d) Diamond is very strong due to the 4 covalent bonds that each of the carbon atoms form. Diamond also has a very high melting point, which means that the cutting tools do not change shape when they become hot when cutting something
3)
a) Carbon
b) 3
c) Hexagons
d) Graphene does conduct electricity because there is one delocalised electron that is free to move throughout the whole structure
4)
a) Weak forces of attraction between the layers
b) It is very easy for the layers to slide over each other
c) Graphite does conduct electricity because there is one delocalised electron that is free to move throughout the whole structure
d) Very high melting and boiling points because graphite is a giant covalent structure that has loads of covalent bonds. These take a lot of energy to break and change state, thus meaning that graphite has very high melting and boiling points
e) Graphite is a good lubricating material as the layers can easily slide over each other
f) Graphite is a good conductor of thermal energy as it has delocalised electrons that carry the thermal energy through the structure/ substance
5) Any 3 from; to cage molecules/ deliver drugs to parts of the body, catalyse reactions, electronics (as they conduct electricity), lubricating materials, increase the strength of materials (whilst not adding much mass/ weight)
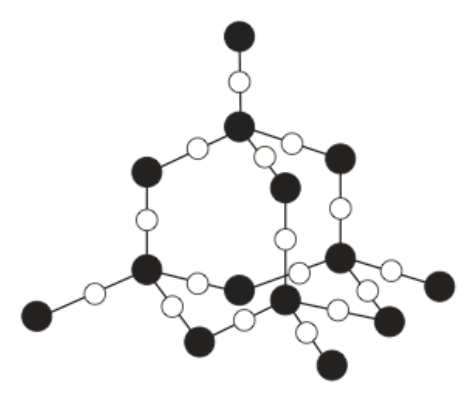
6) Silicon dioxide
a) Diamond
b) Graphene
c) Buckminster/ fullerenes
2)
a) 4
b) Very high melting and boiling points because diamond is a giant covalent structure that has loads of covalent bonds. These take a lot of energy to break and change state, thus meaning that diamond has a very high melting and boiling point
c) Diamond does not conduct electricity as there are no delocalised electrons or ions that can move
d) Diamond is very strong due to the 4 covalent bonds that each of the carbon atoms form. Diamond also has a very high melting point, which means that the cutting tools do not change shape when they become hot when cutting something
3)
a) Carbon
b) 3
c) Hexagons
d) Graphene does conduct electricity because there is one delocalised electron that is free to move throughout the whole structure
4)
a) Weak forces of attraction between the layers
b) It is very easy for the layers to slide over each other
c) Graphite does conduct electricity because there is one delocalised electron that is free to move throughout the whole structure
d) Very high melting and boiling points because graphite is a giant covalent structure that has loads of covalent bonds. These take a lot of energy to break and change state, thus meaning that graphite has very high melting and boiling points
e) Graphite is a good lubricating material as the layers can easily slide over each other
f) Graphite is a good conductor of thermal energy as it has delocalised electrons that carry the thermal energy through the structure/ substance
5) Any 3 from; to cage molecules/ deliver drugs to parts of the body, catalyse reactions, electronics (as they conduct electricity), lubricating materials, increase the strength of materials (whilst not adding much mass/ weight)
6) Silicon dioxide
Questions
1) Name the substance for each of the diagrams below.
1) Name the substance for each of the diagrams below.
2) This question is all about diamond.
a) Diamond is made out of carbon. How many covalent bonds do each of the carbon atoms in diamond form?
b) Does diamond have a very high or a very low melting and boiling point? Explain your answer.
c) Does diamond conduct electricity? Explain your answer.
d) Why is diamond used in cutting tools?
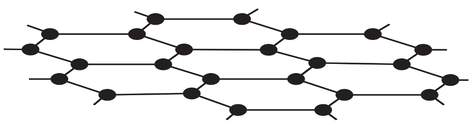
3) The diagram below is of graphene.
a) What element is graphene made from?
b) How many bonds do each of the atoms in graphene form?
c) What is the name of the shape that the atoms in graphene are arranged in?
d) Does graphene conduct electricity? Explain your answer.
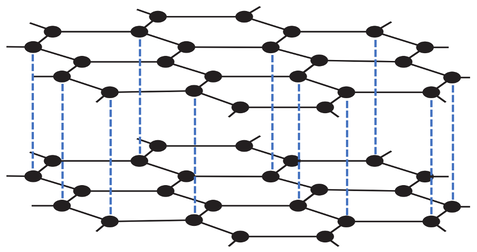
4) The diagram below shows graphite.
b) How many bonds do each of the atoms in graphene form?
c) What is the name of the shape that the atoms in graphene are arranged in?
d) Does graphene conduct electricity? Explain your answer.
4) The diagram below shows graphite.
a) Are there strong or weak forces of attraction between the layers of graphene in graphite?
b) Do the layers easily slide over each other?
c) Does graphite conduct electricity? Explain your answer.
d) Does graphite have a very high or a very low melting and boiling point? Explain your answer.
e) Is graphite a good lubricating material? Explain your answer.
f) Is graphite a good conductor of thermal energy? Explain your answer.
5) Give 3 uses of fullerenes.
6) What is the name of the substance below?
b) Do the layers easily slide over each other?
c) Does graphite conduct electricity? Explain your answer.
d) Does graphite have a very high or a very low melting and boiling point? Explain your answer.
e) Is graphite a good lubricating material? Explain your answer.
f) Is graphite a good conductor of thermal energy? Explain your answer.
5) Give 3 uses of fullerenes.
6) What is the name of the substance below?