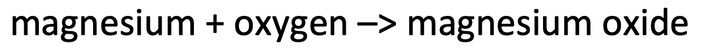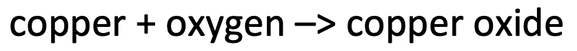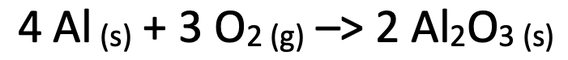C3: Quiz 5 – Answers
1) With chemical reactions, no atoms are created or destroyed. This means that the mass of the reactants will be equal to the mass of the products. Also, there will be the same number of each of the elements in the reactants and the products.
2) 40 grams
3) 8 grams
4) The carbon dioxide produced is a gas and it would escape into the atmosphere as the reaction was not carried out in a closed/ sealed container. This means that when he was measuring the mass after the reaction, he was only measuring the mass of one of the products (calcium oxide; CaO) and not measuring the mass of CO2. Therefore, the mass of the crucible would decrease during the reaction; the mass before the reaction would be greater than the mass after the reaction.
5) Before the reaction, the student was just measuring the mass of the aluminium powder; the oxygen was a gas in the atmosphere, so its mass wasn’t measured. After the reaction, the student will be measuring the mass of the aluminium oxide powder, which will include the mass of the aluminium (already measured) and the mass of oxygen (not already measured as it was in the atmosphere). This means that the mass of the powder after the reaction will be greater than the mass of the powder before the reaction.
6) Yes, the law of conservation of mass still works meaning that the mass of the reactants and products are the same. It is just that the mass of the reactants and products appear to be different because some of the reactants or products are gases and are therefore not included in the mass of the reaction vessel.
1) What is the law of conservation of mass?
2) Magnesium is burnt in air (oxygen) to produce magnesium oxide. The word equation for this reaction is shown below.
3) The word equation below shows the reaction of copper with oxygen to produce copper oxide.
4) A student heated some calcium carbonate in an open crucible. The equation for the reaction is shown below.
5) When aluminium powder is heated, it reacts with oxygen to form aluminium oxide powder. The equation for this reaction is shown below.
6) For the reactions in questions 4 and 5, does the law of conservation of mass still work? Explain your answer.