1) Define pressure.
2) What is the calculation for pressure?
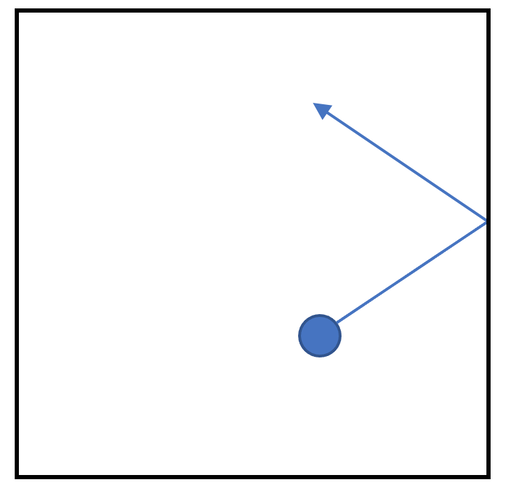
3) The diagram below shows a gas particle hitting the side of a container.
2) What is the calculation for pressure?
3) The diagram below shows a gas particle hitting the side of a container.
When the gas particle hits the side of the container it will exert a force. Draw an arrow on the diagram above to show the direction of this force.
4) We have a container with a fixed volume. If we add more gas particles into the container, what will happen to the pressure? Explain your answer.
5) What would happen to the pressure if we increase the volume of a container and kept the number of gas particles inside the container the same? Explain your answer.
6)
a) When the temperature of gas particles increases, what happens to the energy in the kinetic energy stores of the gas particles? What effect does this have on the average speed of the gas particles?
b)
i) If the volume of a container stayed the same, would an increase in the temperature of the gas particles increase or decrease pressure?
ii) There are two reasons for the change in pressure in part bi; what are these two reasons?
4) We have a container with a fixed volume. If we add more gas particles into the container, what will happen to the pressure? Explain your answer.
5) What would happen to the pressure if we increase the volume of a container and kept the number of gas particles inside the container the same? Explain your answer.
6)
a) When the temperature of gas particles increases, what happens to the energy in the kinetic energy stores of the gas particles? What effect does this have on the average speed of the gas particles?
b)
i) If the volume of a container stayed the same, would an increase in the temperature of the gas particles increase or decrease pressure?
ii) There are two reasons for the change in pressure in part bi; what are these two reasons?