1) Pressure is a force per unit area
2) P = f ÷ a
Pressure = force ÷ area
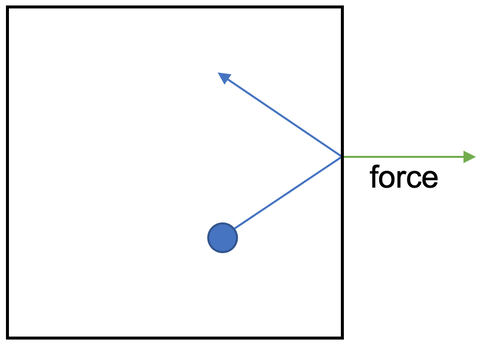
3) An arrow drawn at a right angle to the side of the container – like the green arrow that is shown below
2) P = f ÷ a
Pressure = force ÷ area
3) An arrow drawn at a right angle to the side of the container – like the green arrow that is shown below
4) If we increase the number of gas particles in the container, there would be more gas particles in the same volume. This will result in there being more frequent collisions between the gas particles and the sides of the container, which means that there is a greater force being exerted on the sides of the container. Pressure is force divided by area. So, a greater force and the same area will result in a greater pressure (the area is the same as the container is the same size)
5) If we increase the volume of the container and keep the number of gas particles the same, there will be fewer gas particles in a given volume. This will result in there being less frequent collisions between the gas particles and the sides of the container, which
means that there is a lower force being exerted. Pressure is force divided by area. So, a lower force and a larger area will mean that the pressure in the container will decrease
6)
a) An increase in the temperature of the gas particles increases the energy in the kinetic energy stores of the gas particles. This results in an increase in the average speed of the gas particles
b)
i) Pressure increases
ii) The two reasons are:
- The gas particles will be moving around the container faster as they have more energy in their kinetic energy stores. This means that there will be more frequent collisions between the gas particles and the sides of the container, which increases the overall force on the sides of the container
- The gas particles will be moving around faster, which means that they have a greater momentum (momentum = mass x velocity; a greater velocity means a greater momentum). A greater momentum for the gas particles means that when they collide with the sides of the container, they exert a greater force, which increases the overall force on the sides of the container
Questions
1) Define pressure.
2) What is the calculation for pressure?
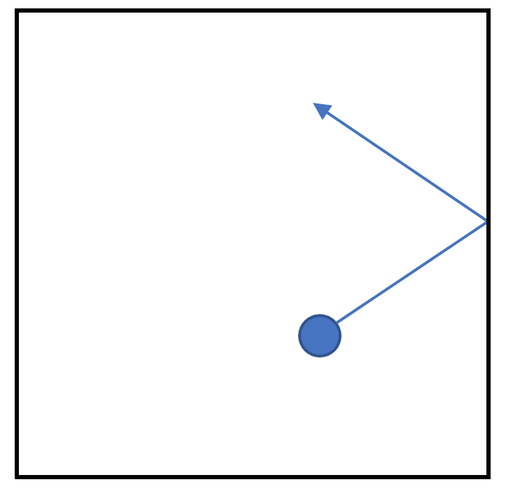
3) The diagram below shows a gas particle hitting the side of a container.
1) Define pressure.
2) What is the calculation for pressure?
3) The diagram below shows a gas particle hitting the side of a container.
When the gas particle hits the side of the container it will exert a force. Draw an arrow on the diagram above to show the direction of this force.
4) We have a container with a fixed volume. If we add more gas particles into the container, what will happen to the pressure? Explain your answer.
5) What would happen to the pressure if we increase the volume of a container and kept the number of gas particles inside the container the same? Explain your answer.
6)
a) When the temperature of gas particles increases, what happens to the energy in the kinetic energy stores of the gas particles? What effect does this have on the average speed of the gas particles?
b)
i) If the volume of a container stayed the same, would an increase in the temperature of the gas particles increase or decrease pressure?
ii) There are two reasons for the change in pressure in part bi; what are these two reasons?
4) We have a container with a fixed volume. If we add more gas particles into the container, what will happen to the pressure? Explain your answer.
5) What would happen to the pressure if we increase the volume of a container and kept the number of gas particles inside the container the same? Explain your answer.
6)
a) When the temperature of gas particles increases, what happens to the energy in the kinetic energy stores of the gas particles? What effect does this have on the average speed of the gas particles?
b)
i) If the volume of a container stayed the same, would an increase in the temperature of the gas particles increase or decrease pressure?
ii) There are two reasons for the change in pressure in part bi; what are these two reasons?