1)
a) What is meant by the term half-life?
b) Is radioactive decay a certain process or a random process?
c) How can we measure the radioactive decay of a substance?
2) We have a sample that contains 800 radioactive isotopes. How many radioactive isotopes will we have after:
a) 1 half-life
b) 2 half-lives
c) 3 half-lives
d) 4 half-lives
e) Will the amount of radioactive decay be the highest during the first half-life or the fourth half-life?
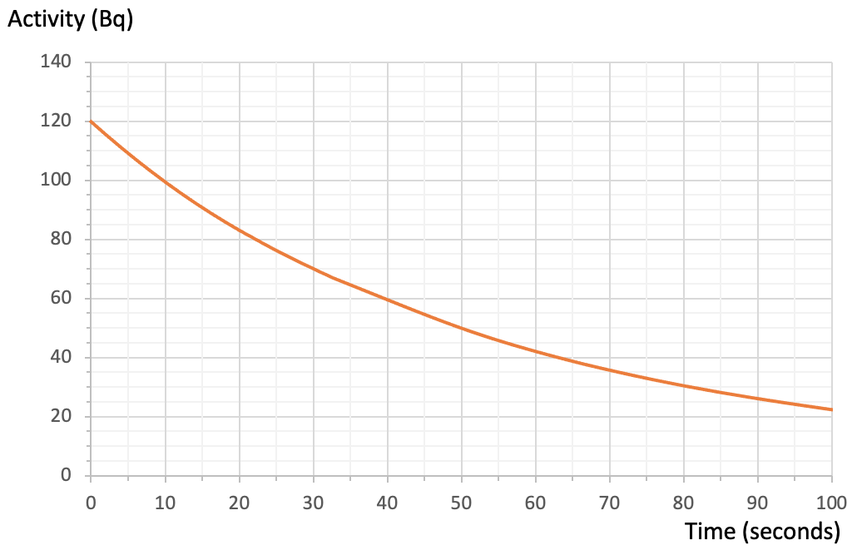
3) The graph below shows how the activity/ count rate for a radioactive isotope varies over time.
a) What is meant by the term half-life?
b) Is radioactive decay a certain process or a random process?
c) How can we measure the radioactive decay of a substance?
2) We have a sample that contains 800 radioactive isotopes. How many radioactive isotopes will we have after:
a) 1 half-life
b) 2 half-lives
c) 3 half-lives
d) 4 half-lives
e) Will the amount of radioactive decay be the highest during the first half-life or the fourth half-life?
3) The graph below shows how the activity/ count rate for a radioactive isotope varies over time.
a) Use the above graph to estimate the time for 1 half-life.
b)
i) How many half-lives will there be after 160 seconds?
ii) Another sample of this substance started with 2,000 radioactive isotopes. How many of the 2,000 radioactive isotopes will remain undecayed after 160 seconds? And, how many of the 2,000 radioactive isotopes will have decayed after the 160 seconds?
4) A sample has an initial activity of 4,000 Bq. The radioactive isotope has a half-life of 30 seconds.
a) What will the activity be after 90 seconds?
b) What will the activity be after 2.5 minutes?
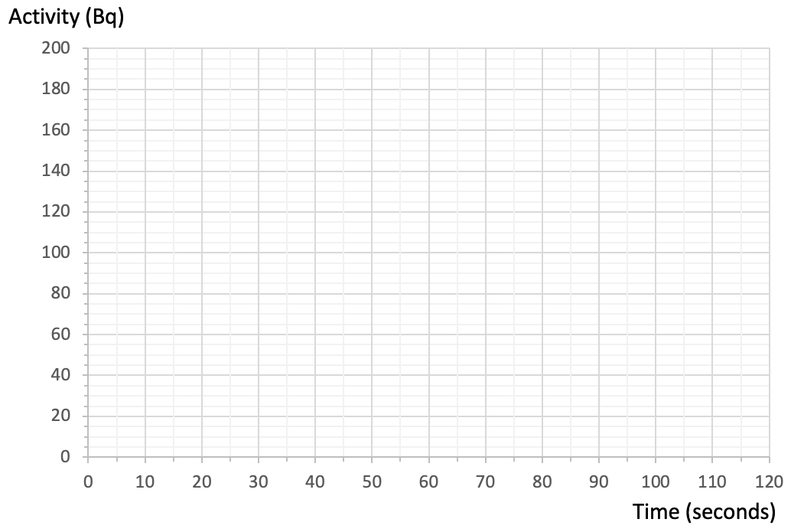
5) The initial activity for a radioactive sample is 180 Bq. The sample has a half-life of 50 seconds.
a) Complete the graph below to show how the activity for a radioactive sample varies over time.
b)
i) How many half-lives will there be after 160 seconds?
ii) Another sample of this substance started with 2,000 radioactive isotopes. How many of the 2,000 radioactive isotopes will remain undecayed after 160 seconds? And, how many of the 2,000 radioactive isotopes will have decayed after the 160 seconds?
4) A sample has an initial activity of 4,000 Bq. The radioactive isotope has a half-life of 30 seconds.
a) What will the activity be after 90 seconds?
b) What will the activity be after 2.5 minutes?
5) The initial activity for a radioactive sample is 180 Bq. The sample has a half-life of 50 seconds.
a) Complete the graph below to show how the activity for a radioactive sample varies over time.
b) Use your graph to predict what the activity will be after 90 seconds.
c) Use your graph to predict the time when the activity will be 70 Bq.
6) The half-life of carbon-14 is 5,700 years. A sample of carbon-14 contains 1,000,000 undecayed nuclei.
a) How many half-lives will have happened after 28,500 years?
b)
i) How many of the 1,000,000 radioactive nuclei will remain undecayed after 28,500 years?
ii) How many of the 1,000,000 radioactive nuclei will have decayed after 28,500 years?
7) We have 2 different radioactive isotopes; isotope A and isotope B. Isotope A has a half-life of 20 days and isotope B has a half-life of 40 years. Both isotopes emit the same type of radiation, and a sample contains an equal number of isotope A and isotope B.
a) Which isotopes will have a higher initial count rate and why?
b) Compare the risks from both of the isotopes after 50 years.
c) Use your graph to predict the time when the activity will be 70 Bq.
6) The half-life of carbon-14 is 5,700 years. A sample of carbon-14 contains 1,000,000 undecayed nuclei.
a) How many half-lives will have happened after 28,500 years?
b)
i) How many of the 1,000,000 radioactive nuclei will remain undecayed after 28,500 years?
ii) How many of the 1,000,000 radioactive nuclei will have decayed after 28,500 years?
7) We have 2 different radioactive isotopes; isotope A and isotope B. Isotope A has a half-life of 20 days and isotope B has a half-life of 40 years. Both isotopes emit the same type of radiation, and a sample contains an equal number of isotope A and isotope B.
a) Which isotopes will have a higher initial count rate and why?
b) Compare the risks from both of the isotopes after 50 years.