1)
a) A half-life is the average time that it takes for the number of radioactive nuclei in a sample to halve
b) Random process
c) We use a Geiger-Muller tube and counter, which counts the number of radioactive particles hitting the counter each second. This is known as the activity or count rate. We measure the count rate in becquerels (Bq) where 1 Bq is 1 decay per second
2)
a) 400
b) 200
c) 100
d) 50
e) Highest during the first half-life
3)
a) 40 seconds
b)
i) 4 half-lives
ii) Undecayed = 125
Decayed = 1,875
4)
a) 500 Bq
b) 125 Bq
5)
a)
a) A half-life is the average time that it takes for the number of radioactive nuclei in a sample to halve
b) Random process
c) We use a Geiger-Muller tube and counter, which counts the number of radioactive particles hitting the counter each second. This is known as the activity or count rate. We measure the count rate in becquerels (Bq) where 1 Bq is 1 decay per second
2)
a) 400
b) 200
c) 100
d) 50
e) Highest during the first half-life
3)
a) 40 seconds
b)
i) 4 half-lives
ii) Undecayed = 125
Decayed = 1,875
4)
a) 500 Bq
b) 125 Bq
5)
a)
The key values to have are:
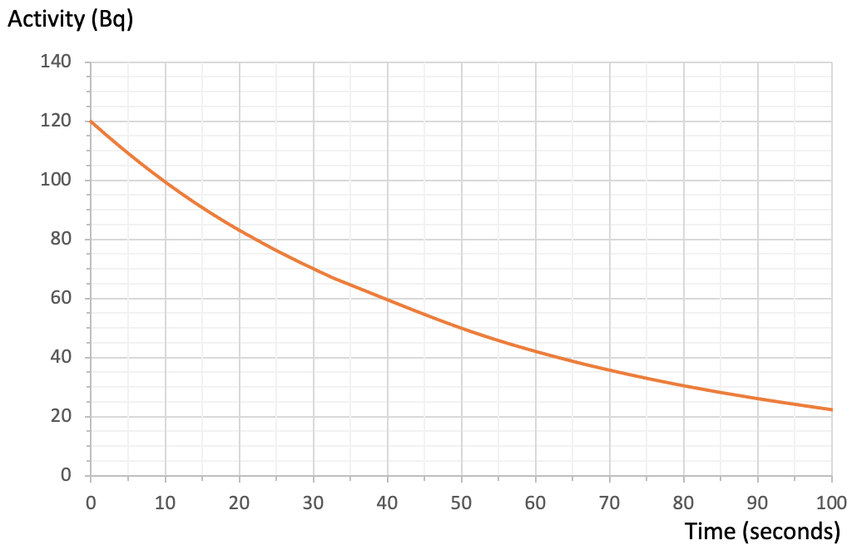
c) 64-72 seconds
6)
a) 5 half-lives
b)
i) 31,250
ii) 968,750
7)
a) Isotope A will have a higher initial count rate as more of the radioactive isotopes will be decaying as isotope A has a shorter half-life (20 days for A compared to 40 years for B)
b) There will be a greater risk from isotope B as there will be more of isotope B in the sample after 50 years as isotope B has a longer half-life (40 years); just over 1 half-life will have happened for isotope B in the 50 years. This means that there will be a lot of isotope B in the sample that will be emitting quite a lot of radiation at the end of 50 years, therefore meaning that there will be a higher risk from isotope B.
There will be a lower risk from isotope A as there will be a smaller quantity of isotope A in the sample after 50 years as isotope A has a short half-life (20 days); lots of half-lives will have happened for isotope A in the 50 years. This means that isotope A will be emitting very little radiation at the end of 50 years, so there is a low risk from isotope A.
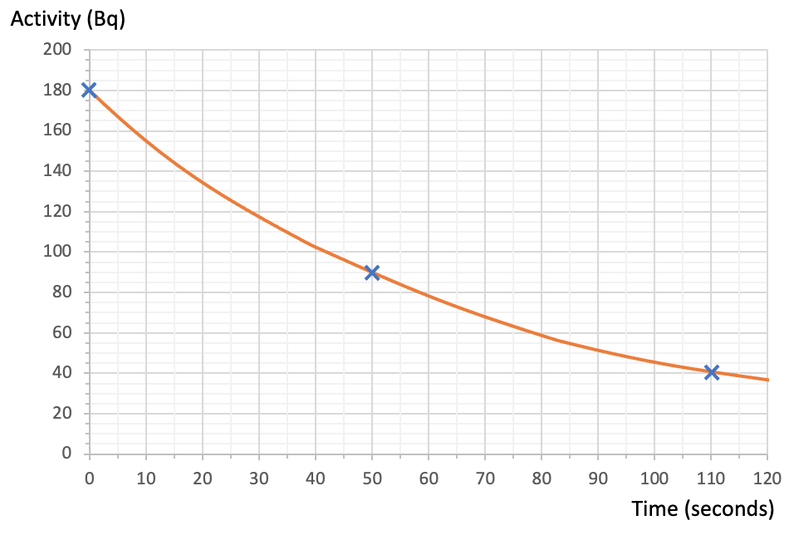
- Activity: 180 Bq, Time: 0 seconds
- Activity: 90 Bq, Time: 50 seconds
- Activity: 45 Bq, Time: 100 seconds
c) 64-72 seconds
6)
a) 5 half-lives
b)
i) 31,250
ii) 968,750
7)
a) Isotope A will have a higher initial count rate as more of the radioactive isotopes will be decaying as isotope A has a shorter half-life (20 days for A compared to 40 years for B)
b) There will be a greater risk from isotope B as there will be more of isotope B in the sample after 50 years as isotope B has a longer half-life (40 years); just over 1 half-life will have happened for isotope B in the 50 years. This means that there will be a lot of isotope B in the sample that will be emitting quite a lot of radiation at the end of 50 years, therefore meaning that there will be a higher risk from isotope B.
There will be a lower risk from isotope A as there will be a smaller quantity of isotope A in the sample after 50 years as isotope A has a short half-life (20 days); lots of half-lives will have happened for isotope A in the 50 years. This means that isotope A will be emitting very little radiation at the end of 50 years, so there is a low risk from isotope A.
Questions
1)
a) What is meant by the term half-life?
b) Is radioactive decay a certain process or a random process?
c) How can we measure the radioactive decay of a substance?
2) We have a sample that contains 800 radioactive isotopes. How many radioactive isotopes will we have after:
a) 1 half-life
b) 2 half-lives
c) 3 half-lives
d) 4 half-lives
e) Will the amount of radioactive decay be the highest during the first half-life or the fourth half-life?
3) The graph below shows how the activity/ count rate for a radioactive isotope varies over time.
1)
a) What is meant by the term half-life?
b) Is radioactive decay a certain process or a random process?
c) How can we measure the radioactive decay of a substance?
2) We have a sample that contains 800 radioactive isotopes. How many radioactive isotopes will we have after:
a) 1 half-life
b) 2 half-lives
c) 3 half-lives
d) 4 half-lives
e) Will the amount of radioactive decay be the highest during the first half-life or the fourth half-life?
3) The graph below shows how the activity/ count rate for a radioactive isotope varies over time.
a) Use the above graph to estimate the time for 1 half-life.
b)
i) How many half-lives will there be after 160 seconds?
ii) Another sample of this substance started with 2,000 radioactive isotopes. How many of the 2,000 radioactive isotopes will remain undecayed after 160 seconds? And how many of the 2,000 radioactive isotopes will have decayed after the 160 seconds?
4) A sample has an initial activity of 4,000 Bq. The radioactive isotope has a half-life of 30 seconds.
a) What will the activity be after 90 seconds?
b) What will the activity be after 2.5 minutes?
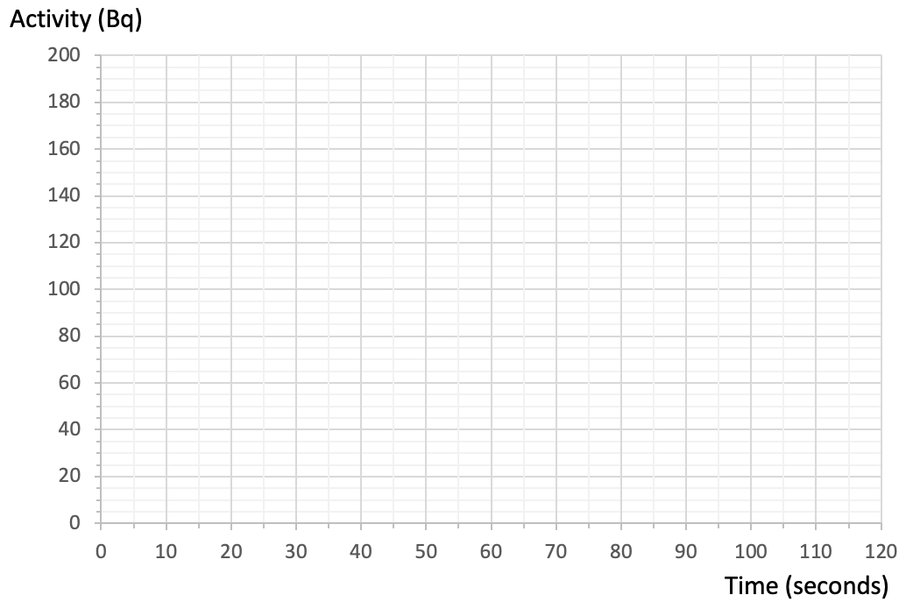
5) The initial activity for a radioactive sample is 180 Bq. The sample has a half-life of 50 seconds.
a) Complete the graph below to show how the activity for a radioactive sample varies over time.
b)
i) How many half-lives will there be after 160 seconds?
ii) Another sample of this substance started with 2,000 radioactive isotopes. How many of the 2,000 radioactive isotopes will remain undecayed after 160 seconds? And how many of the 2,000 radioactive isotopes will have decayed after the 160 seconds?
4) A sample has an initial activity of 4,000 Bq. The radioactive isotope has a half-life of 30 seconds.
a) What will the activity be after 90 seconds?
b) What will the activity be after 2.5 minutes?
5) The initial activity for a radioactive sample is 180 Bq. The sample has a half-life of 50 seconds.
a) Complete the graph below to show how the activity for a radioactive sample varies over time.
b) Use your graph to predict what the activity will be after 90 seconds.
c) Use your graph to predict the time when the activity will be 70 Bq.
6) The half-life of carbon-14 is 5,700 years. A sample of carbon-14 contains 1,000,000 undecayed nuclei.
a) How many half-lives will have happened after 28,500 years?
b)
i) How many of the 1,000,000 radioactive nuclei will remain undecayed after 28,500 years?
ii) How many of the 1,000,000 radioactive nuclei will have decayed after 28,500 years?
7) We have 2 different radioactive isotopes; isotope A and isotope B. Isotope A has a half-life of 20 days and isotope B has a half-life of 40 years. Both isotopes emit the same type of radiation, and a sample contains an equal number of isotope A and isotope B.
a) Which isotopes will have a higher initial count rate and why?
b) Compare the risks from both of the isotopes after 50 years.
c) Use your graph to predict the time when the activity will be 70 Bq.
6) The half-life of carbon-14 is 5,700 years. A sample of carbon-14 contains 1,000,000 undecayed nuclei.
a) How many half-lives will have happened after 28,500 years?
b)
i) How many of the 1,000,000 radioactive nuclei will remain undecayed after 28,500 years?
ii) How many of the 1,000,000 radioactive nuclei will have decayed after 28,500 years?
7) We have 2 different radioactive isotopes; isotope A and isotope B. Isotope A has a half-life of 20 days and isotope B has a half-life of 40 years. Both isotopes emit the same type of radiation, and a sample contains an equal number of isotope A and isotope B.
a) Which isotopes will have a higher initial count rate and why?
b) Compare the risks from both of the isotopes after 50 years.